Bone Biology

Bone healing depends on biological factors and the mechanical conditions in the defect region. Despite the advances in fracture fixation, there remains a subset of patients that suffer from healing complications, resulting in delayed healing and non-unions. Currently it is not possible to reliably identify healing complications at an early stage when treatments may be more effective. Within the Bone Biology Focus Area, we study biological factors involved in the different phases of bone healing with a major focus on early immunological, angiogenic and mechano-molecular components.
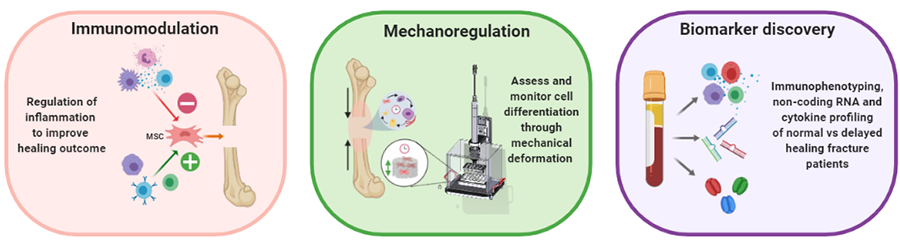
The immune system is involved in guiding and directing the healing response. We are investigating how modulation of inflammation may be used to enhance the bone healing process, as well as assessing the potential of immune cell characterization to be used as a predictive biomarker of the individual healing potential.
Angiogenesis is essential for bone healing. We are investigating autologous sources of proangiogenic cells and stimuli for the understanding and the promotion of an adequate vascular environment in injured bone tissue using both in vitro (bioreactors, microfluidic systems) and in vivo (contrast agent, time-lapsed micro-CT) approaches.
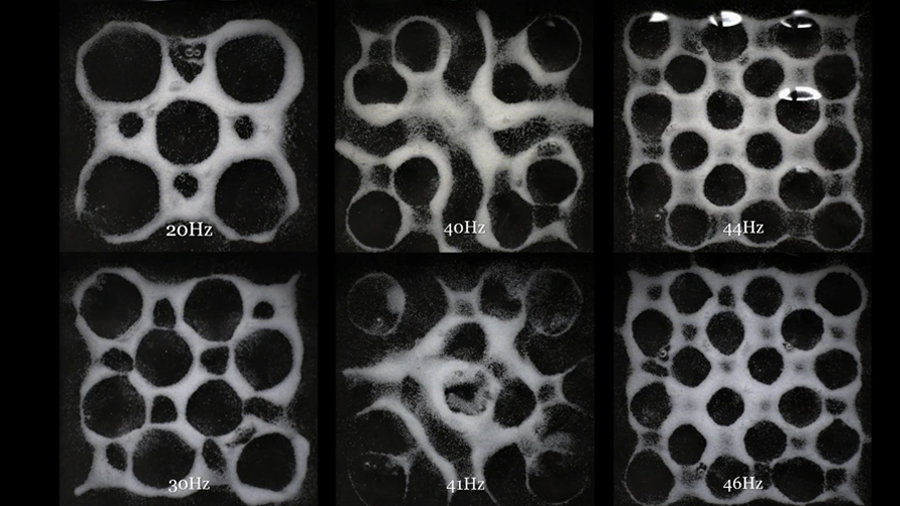
Mechano-molecular mechanisms are important for successful bone healing. Via our novel technology we want to precisely study how mechanics influence molecular mechanism during bone healing in vivo (femur defect loading model in mice) and in vitro (bone bioreactor). In combination with emerging molecular omics techniques, we want to comprehensively characterize the local and systemic mechano-molecular regulation of bone healing. Via this combined in vivo and in vitro approach, we aim for identification of novel therapeutic targets, systemic biomarkers, and mechanical intervention therapies relevant towards translation of personalized medicine approaches for impaired healing conditions.