Bio-ECM offers a safe and effective, treatment option for bone growth and bridging of bone defects

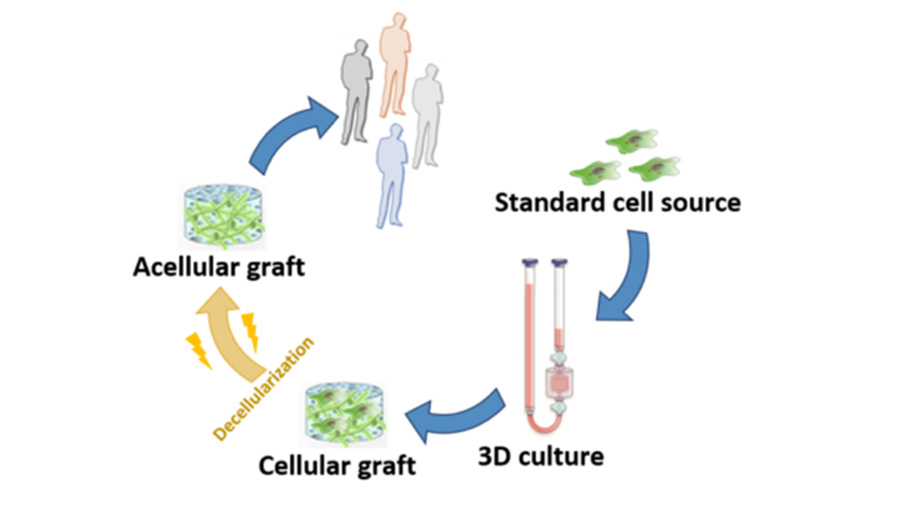
Fig 1. BioECM vision of futuristic production line for engineered biological tissues
A tissue-engineered, osteoinductive bone graft substitute—ten years in development by scientists in Sweden and Switzerland—is poised to provide surgeons with a treatment option which combines the safety of autograft with the efficacy and reproducibility of synthetic osteoinductive products, thereby maximizing patient benefits. The Bio-ECM project, which seeks to develop and prove efficacy of an engineered decellularized human extracellular matrix (ECM), is supported by AO development incubator resources made available through the AO Innovation Translation Center (AO ITC) Technology Transfer.
Led by Prof Paul Bourgine of Lund University, Sweden, and Prof Ivan Martin, Department of Biomedicine, University of Basel, Switzerland, and executed by their respective teams , the Bio-ECM project represents a safe, osteoinductive alternative or adjunct to autografts which are the current gold standard for bone grafting.
Alternative to the gold standard
Autografts involve harvesting and transplanting a patient's own bone graft to the surgical site; this osteogenic method results in consistent bone fusion, no immune reaction, and is the most natural and straightforward way for treating bone defects. However, this method also presents significant drawbacks, including limited material availability, the potential for chronic pain at the harvesting site, and variable quality of the material, which is dependent on the patient's health status. These limitations have given rise to a need for alternatives—a need that Bourgine and Martin set out to address more than a decade ago.
The Bio-ECM project addresses those limitations by engineering in the lab human tissue grafts, enriched with bone morphogenetic protein 2 (BMP-2) to enhance its bone-forming properties. Instrumental to the project is the use of a unique human cell line, ensuring standardized and limitless production of Bio-ECM. One key feature is that, unlike most grafts and materials that mimic bone structure and composition, Bio-ECM consist in a cartilage matrix, instructing bone formation through endochondral ossification: the body's natural bone forming and repairing process. Most remarkably, the necessary cells exploited for tissue formation are ultimately removed; the Bio-ECM is thus a cell-free implant, instructing bone repair in patients.
Efficacy of repair, safety, the natural bone-forming process as well as reproducibility are Bio-ECM hallmarks. In animal models at ectopic implantation sites, the grafts resulted in consistent and rapid formation of naturally structured bone composed of a cortical shell and a cancellous inner structure. From a safety perspective, the Bio-ECM relies on a physiological cocktail of factors in contrast to methods using very high doses of a single BMP, which can promote inflammation, cancer, or result in overshooting bone growth. Since Bio-ECM consist in a cell-free tissue graft, it can be easily stored and made readily available, as an off the shelf material, and efficient treatment option.
Spinal fusion procedures are one primary indication where Bio-ECM could transform patient outcomes. It bears the potential to be used as a filler for interbody fusion spacers or posterior interbody fusion, as well as in cervical fusion.
Support beyond funding
Bourgine said the AO wasn’t new to him when he and Martin approached the AO ITC Technology Transfer for support: a portion of the Bio-ECM research had been supported by a 2015 AO Start-Up Grant.
“Finding resources to sustain the fundamental development of a project is always challenging, but what is unique about the AO ITC Technology Transfer is the support beyond funding. This includes expertise, support for managing the project, and—importantly—access to the AO’s network of surgeons,” he explains. “We also appreciate the Technology Transfer’s collaborative approach, which has really driven the success of our project.”
Bio-ECM has received positive feedback from AO Trauma, AO Spine, and AO CMF surgeons, and has cross-divisional potential, such as for treatment of nonunion of long bone fractures, critical size defects in long bones, and mandibular defects.
Moving ahead
With development incubator support over the past two years, the Bio-ECM project has reached several milestones, including filing a patent application and establishing Freedom to Operate (FTO)—key steps to industrialization; achieving proof of concept in a small animal model; identifying, establishing, and testing GMP-approved manufacturing components; and confirming the regulatory classification and pathway for the material with a national Competent Authority. Still ahead are the planning and execution of a large-animal study to provide further proof of safety and efficacy of the material in a functional model. Further meetings with Competent Authorities will also be required to ultimately bring Bio-ECM to its first clinical application and make it available to the surgeon community for the patients' benefit.