miRNA analysis to discover fracture related biomarkers
Background
Biomarkers predictive of fracture healing outcomes would provide a useful tool to allow surgeons to proactively make patient based clinical decisions. Currently, even in high risk groups, there are no accurate ways to determine the potential of a particular patient to progress to delayed or non-union. Such a tool would enable more reliable patient stratification, thus allowing for earlier diagnosis and increasing the potential success of additional early interventions by the surgeon. Small non-coding RNA sequences, such as microRNA (miRNA) have been shown to be powerful regulators of cellular behavior in both healthy and diseased environments. They can function by interacting with messenger RNA sequences and thereby modifying protein expression. miRNAs normally act intracellularly, but due to the action of extracellular vesicles (EVs) released by cells they are able to signal over large distances and thus EVs are a critical signaling pathway between different cells. EVs and miRNA have the advantage of being extremely stable, detectable in complex body fluids such as serum, and provide information directly relating to cellular function. MicroRNA (miRNA) studies are already transitioning from basic research applications to clinical applications in areas such as cancer diagnosis.
Goal
Within this project we aim to identify fracture related non coding RNA sequences, present in extracellular vesicles, in the serum of patients. Then establish their function within primary human mesenchymal stem cells and propose predictive markers that could be used to screen patients early after injury. In addition, functionally active miRNA species identified as lacking in non-healing patients can also be used as a potential off-the-shelf treatment to enhance fracture repair in patients shown to have a decreased level of expression.
Results
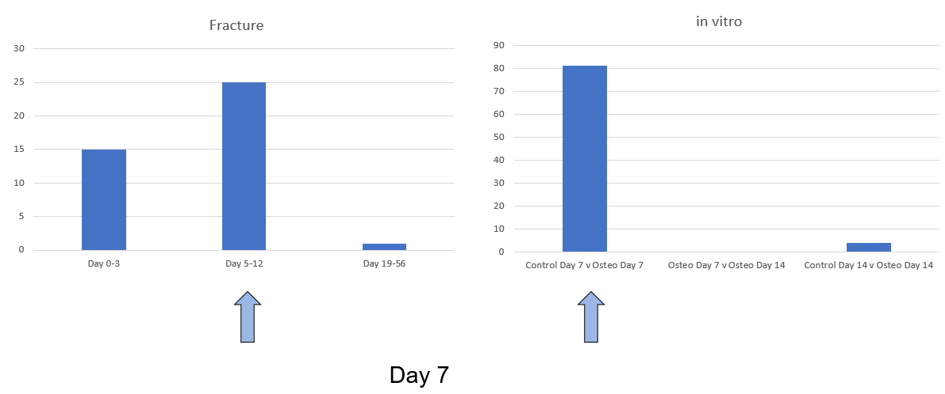
Serum was obtained from patients at various times after injury day 0-3, day 5-12 and day 19-56 and whole non-coding RNA was sequenced. As a comparator, mesenchymal stromal cells (MSCs) were induced to undergo osteogenesis and compared to unstimulated controls. We have identified several miRNA targets that are regulated during early osteogenesis. We also identified the presence of some of these markers in serum taken from fracture patients within days after fracture (Figure). Furthermore, we are also one of the first groups to identify piRNA and circular RNA markers of osteogenesis. This work will be continued in a new follow up project.
-
Publication
Della Bella E, Stoddart M. Cell detachment rapidly induces changes in non-coding RNA expression in human mesenchymal stromal cells. Biotechniques. 2019 Oct 17. doi: 10.2144/btn-2019-0038.
Kadekar S, Barbe L, Stoddart M, Varghese OP, Tenje M, Mestres G. Effect of the Addition Frequency of 5-Azacytidine in Both Micro- and Macroscale Cultures. Cell Mol Bioeng. 2020;epub Oct 06.
Della Bella E, Koch J, Baerenfaller K. Translation and emerging functions of non-coding RNAs in inflammation and immunity. Allergy. 2022;epub Jan 29. https://doi.org/10.1111/all.15234Isenmann M, Stoddart MJ, Schmelzeisen H, Gross C, Della Bella E, Rothweiler RM. Basic principles of RNA interference: Nucleic acid types and in vitro intracellular delivery methods. Micromachines. 2023;14(7). https://doi.org/10.3390/mi14071321
-
Presentation
Della Bella E, Ahmad P, Buetti-Dinh A, Licandro G, Basoli V, Alini M, Stoddart M. Dexamethasone: A friend or a foe in osteogenic differentiation of human BMSCS?
2021 TERMIS world congress digital (poster)Della Bella E, Menzel U, Basoli V, Tourbier C, Alini M, Stoddart MJ. Dexamethasone regulates circular RNA expression during human bone marrow mesenchymal stromal cells differentiation. 2021 TERMIS world congress digital (poster)
Breulmann FL, Ramasamy S, Herzog M, Pandian GN, Della Bella E, Stoddart MJ. Differentially expressed microRNAs during early endochondral differentiation of human mesenchymal stromal cells as biomarkers for non-union fractures. 2022 SSB+RM (poster)
Stoddart MJ. In vitro testing of bone biomaterials - opportunities and challenges. 2022 TERMIS EU (oral / invited Keynote)Breulmann F, Herzog M, Stoddart M, Della Bella E. Differentially expressed microRNAs during endochondral differentiation of human bone marrow derived mesenchymal stromal cells to identify possible biomarkers for non-union fractures. 2022 TERMIS EU (oral)
-
PartnerKubosch J (MD), Albert-Ludwigs University Medical Center Freiburg, Germany