local-delivery-of-il-1ra-as-a-strategy-to-enhance-long-bone-healing
Background
Although 90% of fractures typically heal without complications, there remains a small proportion (≤ 10%) of fractures that experience delayed healing or non-union. In patients with such healing complications, there appears to be an important contribution of an inappropriately maintained pro-inflammatory environment to the defective fracture healing process. Thus, immunomodulation of the local fracture microenvironment, such as by enhancing anti-inflammatory cytokine production, could be an effective way to enhance fracture healing in troublesome healing environments. This project focuses on investigating the therapeutic efficacy of IL-1Ra, the receptor antagonist of the potent pro-inflammatory cytokine IL-1β.
Goal
The goals of this project include the identification and characterization of a suitable pro-inflammatory endochondral bone healing model, and to test the efficacy of IL-1Ra delivered locally to the site of injury to promote bone healing.
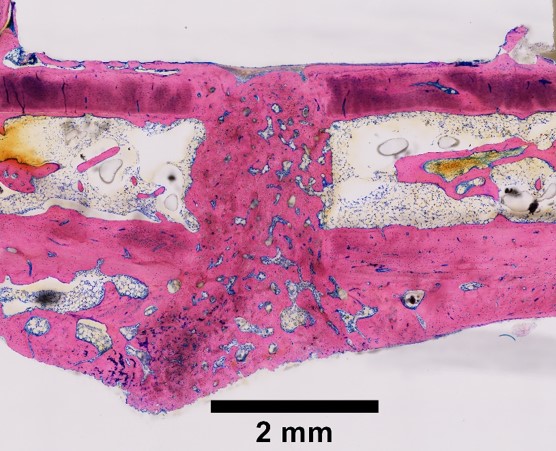
Results
A 2 mm femoral defect was created in skeletally mature female Fischer 344 rats, following internal plate fixation using a customized 1.25 mm-thick PEEK plate. This model of non-union has been characterized based on microCT and histological assessment, with robust bone formation but an ultimate failure to bridging up to after 14 weeks post-surgery. The creation of the defect led to elevated circulating levels of IL-1β, which were maintained up to 10 days after surgery. Characterization of the early and local inflammatory response to the defect demonstrated there was a time-dependent shift in the ratio between IL-1β and IL-1Ra. Our results show that IL-1β protein levels at the defect site persisted up to 7 days post-surgery, whilst IL-1Ra markedly increased from 10 days onwards (p<0.01). Additionally, we have shown that up to 16 different inflammatory cytokines and chemokines in the bone marrow of the femur undergo significant changes in expression within 14 days post-surgery. Taken together, our findings suggest that, despite the small size of the defect, this model can be characterized by non-union in the long-term (14 weeks), and a persistent inflammatory response in the short-term (<2 weeks). With these characteristics, this model represents an excellent platform to determine the therapeutic efficacy of immunomodulatory strategies, including local delivery of IL-1Ra. On-going studies are investigating the efficacy of different biomaterials for delivery of recombinant proteins, including IL-1Ra and BMP-2, as a means of enhancing bone formation.
-
PublicationLackington WA, Gomez-Sierra MA, González-Vázquez A, O’Brien FJ, Stoddart MJ, Thompson K. Non-viral Gene Delivery of Interleukin-1 Receptor Antagonist Using Collagen-Hydroxyapatite Scaffold Protects Rat BM-MSCs from IL-1β-Mediated Inhibition of Osteogenesis. Front Bioeng Biotechnol. 2020;8:1180
Lackington WA, Gehweiler D, Zderic I, Nehrbass D, Zeiter S, González-Vázquez A, O’Brien FJ, Stoddart MJ, Thompson K. Incorporation of hydroxyapatite into collagen scaffolds enhances the therapeutic efficacy of rhBMP-2 in a weight-bearing femoral defect model. Mater Today Commun. 2021;29:102933. https://doi.org/10.1016/j.mtcomm.2021.102933
Lackington WA, Gehweiler D, Zhao E, Zderic I, Nehrbass D, Zeiter S, Gonzalez-Vazquez A, O'Brien FJ, Stoddart MJ, Thompson K. Interleukin-1 receptor antagonist enhances the therapeutic efficacy of a low dose of rhBMP-2 in a weight-bearing rat femoral defect model. Acta Biomater. 2022;epub Jul 12. https://doi.org/10.1016/j.actbio.2022.07.012
-
Presentation
Lackington WA, et al., Local administration of IL-1Ra as a strategy to enhance long bone healing. American Society for Bone and Mineral Research Annual Meeting, Orange County, Orlando, Florida, US, 2019.
Gomez MA, et al., Local non-viral gene delivery to immunomodulate and enhance fracture healing. Annual Meeting of the European Orthopaedic Research Society, Maastricht, the Netherlands, 2019.
Lackington WA, et al., Local delivery of interleukin-1 receptor antagonist (IL-1Ra) to enhance long bone healing. 10th Tissue Engineering and Regenerative Medicine International Society (TERMIS) EU Chapter Meeting, Rhodes, Greece, 2019.
Lackington WA, et al., Local delivery of interleukin-1 receptor antagonist (IL-1Ra) to enhance long bone healing. 6th Swiss Bone and Mineral Society Annual Meeting, Bern, Switzerland, 2019.
Lackington W, Gomez M, Vazquez A, O'Brien FJ, Thompson K. Local non-viral gene delivery to immunomodulate and enhance fracture healing. 2020 GR forscht virtual (oral).
Stoddart M. Mechanisms of bone regeneration. 2021 WIRM (oral)
Lackington WA, Gehweiler D, Zderic I, Nehrbass D, Zeiter S, González-Vázquez A, O'Brien F, Stoddart M, Thompson K. IL-1 receptor antagonist enhances rhBMP-2-induced new bone formation during femoral fracture healing in rats. 2021 ES Biomat virtual (oral)
Lackington WA, Gehweiler D, Zderic I, Nehrbass D, Zeiter S, González-Vázquez A, O'Brien FJ, Stoddart MJ, Thompson K. IL-1Ra enhances rhBMP-2-induced new bone formation during femoral fracture healing. 2021 eCM cancelled (poster)/abstract published. https://www.ecmconferences.org/abstracts/2021/Collection3/poster.pdf
Kuhn E, Hofstee MI, Chittò M, Moriarty TF. Antibiotic evasion by Staphylococcus aureus abscess communities in vitro. 2023 YSS SSB+RM (poster)