Exopolysaccharide coated material surfaces modulating fracture immune status and enhancing bone healing
Background
One of the most challenging complications in trauma surgery is fracture related infection (FRI). In chronically infected non-unions, treatment always includes extensive debridement to remove necrotic and infected bone, often resulting in large defects requiring elaborate and prolonged bone reconstruction techniques. There is a perception in the clinical community that in the patients with a chronically infected non-union compared to patients requiring acute resection due to trauma or osteosarcoma (non-infected) poorer repair is achieved. In patients, it is known that compromised immune status can delay bone healing. In experimental studies it was seen that bone repair can be compromised in animals previously infected compared with non-infected equivalents. In trauma patient treated for infection, it is likely that the immune status of the wound is such that the normal healing cascade is deregulated, leading to slower and suboptimal recovery. Therefore, reinstalling a normal or pro-healing immune status after treatment of a bone infection may positively impact the speed of bone healing.
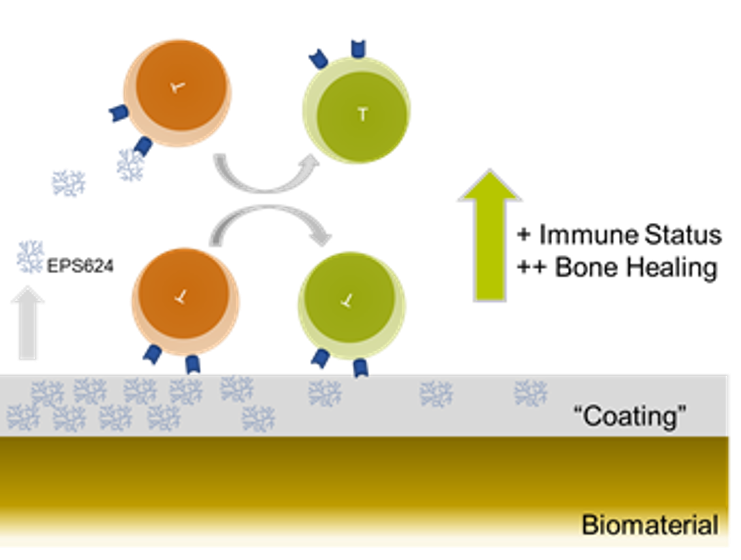
In this project, we aim to reinstall an immunocompetent fracture milieu via the locally delivery (implant coating) of an immune-modulating exopolysaccharide (EPS) secreted by commensal microbe Bifidobacterium which has shown to increase anti-inflammatory (immunoregulatory) cytokine secretion, which we propose will reduce the pro-inflammatory environment present in an infected wound (Figure).
-
Publication
Bagnol R, Grijpma D, Eglin D, Moriarty TF. The production and application of bacterial exopolysaccharides as biomaterials for bone regeneration. Carbohydr Polym. 2022;291:119550. https://doi.org/10.1016/j.carbpol.2022.119550
-
PresentationBagnol R, Sprecher CM, Peroglio M, Richards RG, Eglin D. 3D printing of a calcium phosphate paste for the production of patient specific bone substitutes. 2020 GR forscht virtual (oral).
Bagnol R, O'Mahony L, Moriarty TF, Eglin D. Physico-chemical characterization of polyelectrolyte coatings composed of a bacterial exopolysaccharide and chitosan. 2022 GR forscht (oral)Bagnol R, O'Mahony L, Moriarty TF, Eglin D. Physico-chemical and immunoregulatory characterization of polyelectrolytes coatings from a bacterial exopolysaccharide. 2022 ESB (Biomaterials) (poster)
Bagnol R, Siverino C, Barnier V, O'Mahony L, Grijpma D, Eglin D, Moriarty TF. Physico-chemical characterization an immunomodulatory bacterial exopolysaccharide polyelectrolyte coating.
2023 SfB (poster)Bagnol R, Siverino C, Barnier V, O'Mahony L, Grijpma D, Eglin D, Moriarty TF. Physico-chemical and biological characterization of polyelectrolyte coatings with immunoregulatory properties derived from a Bifidobacterium longum exopolysaccharide. 2023 ESB (Biomaterials) / (oral)