Bottom up printing approach 2 (BUPA 2)
Background
Mandibular defects are due to trauma, tumor, infection or malformation. Surgical reconstruction can be challenging and includes a variety of surgical techniques. A bone graft (vascularized or not) is harvested from different skeletal regions and transferred from the donor site to the recipient site. Current bone grafting procedures have several disadvantages, the main ones being restricted availability, donor site morbidity and limitations in reproducing the three-dimensional (3D) structure of the mandible.
3D printing of large scaffolds to replace hard tissues offers great promise for regeneration of large bone defects. Current approaches aim to produce single patient-specific implants. While this leads to ease of handling it raises questions such as how cells and blood vessels can penetrate large initially acellular constructs.
Goal
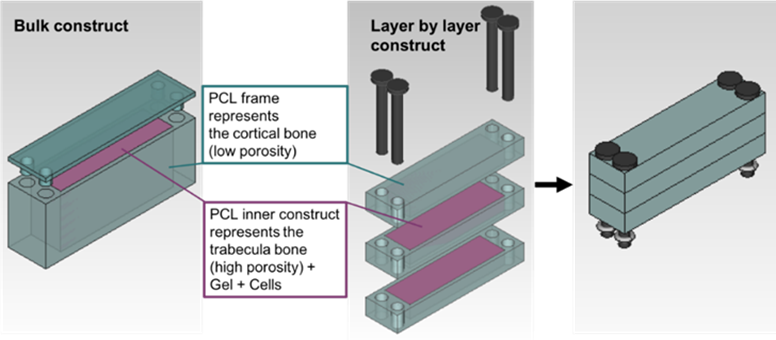
The objective of the present project is to develop and evaluate 3D printed multi-component scaffolds for mandibular defect reconstruction that can be seeded with osteoprogenitor cells and assembled intraoperatively. We will use a standard computed tomography (CT) protocol and computer-assisted planning to fabricate individualized, multi-component scaffold templates. Physical scaffold models will be then manufactured via 3D printing. Unlike classical 3D printed scaffolds, the final scaffold will be printed as multiple interlocking pieces, that can be specifically seeded with different cell types and assembled like building blocks (= Bottom up printing approach) intraoperatively. Cell distribution and viability will be compared between a bottom up layered scaffold and a classical top down single scaffold of the same dimensions.
With this approach we aim to define a new paradigm for regeneration of large bone defects and reconstruction of mandible 3D structure while avoiding the disadvantages associated with traditional bone grafting procedures.
-
Publication
Stoddart M, Richards RG. Growth factors for craniomaxillofacial applications. In: Ehrenfeld M, Futran ND, Manson PN, Prein J (editors). Advanced Craniomaxillofacial Surgery. Tumor, Corrective Bone Surgery and Trauma. Davos: AO Foundation / Thieme; 2020. p. 68-79.
Ahmad P, Della Bella E, Stoddart MJ. Applications of Bone Morphogenetic Proteins in Dentistry: A Bibliometric Analysis. Biomed Res Int. 2020;2020:5971268.
Ahmad P, Arshad AI, Della Bella E, Khurshid Z, Stoddart M. Systemic Manifestations of the Periodontal Disease: A Bibliometric Review. Molecules. 2020;25:4508.Hasler J, Hatt LP, Stoddart MJ, Armiento AR. Stable reference genes for qPCR analysis in BM-MSCs undergoing osteogenic differentiation within 3D hyaluronan-based hydrogels. Int J Mol Sci. 2020;21(23):9195.
Hasler J. Identification of a suitable RNA extraction method and stable reference genes for qRT-PCR in 3D osteogenic cultures of mesenchymal stromal cells.
2020 ETH Zürich (MJ Stoddart, AR Armiento, SJ Ferguson) - MSc ETH HSTAhmad P, Stoddart MJ, Della Bella E. The role of noncoding RNAs in osteogenic differentiation of human periodontal ligament stem cells. CMTRO. 2021;6:2472751221999229. https://doi.org/10.1177/2472751221999229
Ahmad P, Alam MK, Aldajani A, Alahmari A, Alanazi A, Stoddart M, Sghaireen MG. Dental robotics: A disruptive technology. Sensors. 2021;21(10):3308. https://doi.org/10.3390/s21103308
Breulmann FL, Hatt LP, Schmitz B, Wehrle E, Richards RG, Della Bella E, Stoddart MJ. Prognostic and therapeutic potential of microRNAs for fracture healing processes and non-union fractures: A systematic review. Clin Transl Med. 2023;13(1):e1161. https://doi.org/10.1002/ctm2.1161
-
Presentation
Hatt LP, Wirth S, Thompson K, Mys K, Gehweiler D, Eglin D, Stoddart MJ, Armiento AR. LEGO®-inspired microporous scaffolds for personalised mandibular bone repair. 2021 TERMIS world congress digital (oral)
Hatt LP, Wirth S, Ristaniemi A, Thompson K, Eglin D, Stoddart MJ, Armiento AR. LEGO®-inspired microporous scaffolds for personalised mandibular bone repair. 2021 eCM cancelled (oral)/abstract published. https://www.ecmconferences.org/abstracts/2021/Collection3/oral.pdf
Hatt LP, Wirth S, Armiento AR, Ristaniemi A, Thompson K, Gehweiler D, Eglin D, Stoddart MJ. Delivery of mesenchymal stromal cells using collagen membranes embedded in LEGO®-inspired multicomponent scaffolds for personalised mandibular defect repair. 2022 SSB+RM (oral)
Hatt LP, Armiento AR, Pirera ME, Stoddart MJ. β-TCP from 3D-printed scaffold can act as an effective phosphate source during the osteogenic differentiation of human mesenchymal stromal cell. 2023 YSS SSB+RM (poster)
van der Heide D, Della Bella E, Yuan H, de Groot-Barrère F, Stoddart MJ, D'Este M. 3D-printed bone mimicking scaffolds composed of hyaluronan, collagen and calcium phosphate particles for bone regeneration. 2023 ORS (poster)