Assessing and rectifying donor variation for musculoskeletal applications (Varidon)
Background
In the development of cell-based therapies for osteochondral defects and diseases there is still considerable debate regarding which is the most suitable source of cells. A significant amount of work has been performed to investigate the suitability of bone marrow derived mesenchymal stromal cells (BMSCs). While showing promise, studies involving primary human MSCs suffer from the wide donor variation observed. For in vitro studies this leads to challenges involving statistical significance and the requirement for repeats from multiple donors. For clinical translation of autologous therapies, the lack of an underlying mechanism causing the variation would result in a population of poor responders being unsuitable for autologous cellular based therapies. However, the prediction of which patients are likely to respond well is currently not possible and the spread of responses obtained is loosely dismissed as donor variation. If the underlying functional mechanism for the failure to respond was determined, not only would the suitability of a particular donor for cell therapy be able to be predicted, but also a corrective measure may be realized.
Goal
Within this study we aim to identify predictive markers of human MSC chondrogenesis that can be used to stratify patient populations.
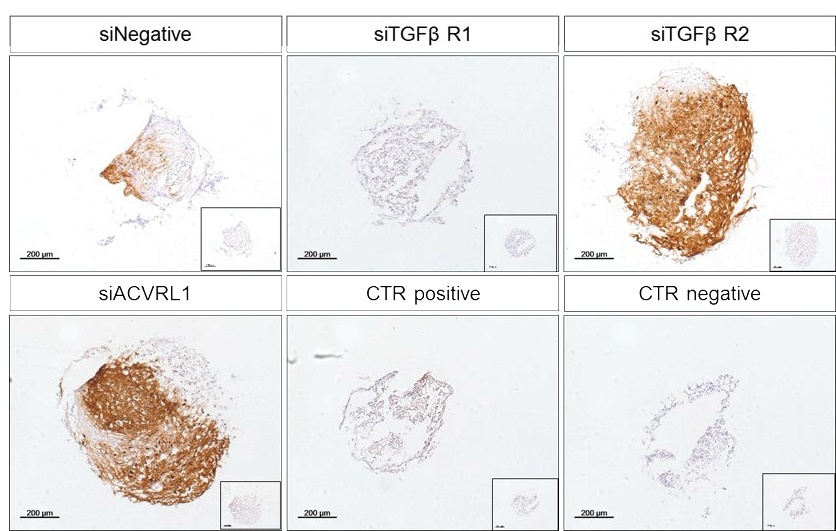
Results
We have identified a prospective chondroprogenitor marker profile that will allow for better patient stratification. Using this profile, we are also able to reverse the deficit, thus converting poor responders to more chondrogenic cells by way of a simple treatment with siRNA silencing. We are further exploring alternative strategies to manipulate cell function, using the profile as an outcome parameter. The ability to reverse this functional deficit will open new avenues for further cartilage repair therapies.
-
Publication
Herrmann M, Hildebrand M, Menzel U, Fahy N, Alini M, Lang S, Benneker L, Verrier S, Stoddart MJ, Bara JJ. Phenotypic Characterization of Bone Marrow Mononuclear Cells and Derived Stromal Cell Populations from Human Iliac Crest, Vertebral Body and Femoral Head. Int J Mol Sci. 2019 Jul 14;20(14). pii: E3454. doi: 10.3390/ijms20143454.
Graceffa V, Vinatier C, Guicheux J, Stoddart M, Alini M, Zeugolis DI. Chasing Chimeras – The elusive stable chondrogenic phenotype. Biomaterials. 2019;192:199-225
Method of determining or influencing the chondrogenic potential of mesenchymal stromal cells Stoddart M, Alini M (patent submitted)
-
Presentation
Stoddart MJ. Stem Cells: What is the real science? ICRS 2019, Vancouver, Canada. (Plenary Speaker.)
Basoli V, Della Bella E, Alini M, Stoddart MJ. Role of dexamethasone and (+)-ZK 216348 during chondrogenic fate in bone marrow stem cells. 2019 TERMIS EU (oral)
Della Bella E, Basoli V, Alini M, Stoddart MJ. Dexamethasone drives early osteogenic differentiation by modulation of SOX9 and PPARG expression. 2019 TERMIS EU (oral)
-
Partner
Johnstone B (Prof), Oregon Health & Science University, USA