A novel concept for guided growth regulation (GoForce)
Background
Corrections of limb deformities, in particular varus-valgus and leg length discrepancies are frequent interventions in pediatric orthopedic surgery. Depending on the severity, deformities can be balanced by temporary epiphysiodesis, where the growth is guided by blocking the physis. Currently utilized implants have their disadvantages, being not 'passively' safe and requiring timely surgical removal, as sustained growth leads to steady rise of the implant reaction force, resulting in such devastating events as implant-related failures or unwanted permanent physeal growth plate closure due to over-excessive forces. A novel 'passively' safe implant concept was developed applying growth independent constant compression force to the physis, hypothesized to provide safe, reliable and controllable treatment.
Goal
To test the functionality and efficacy of the proposed implant concept in a large animal model.
Results
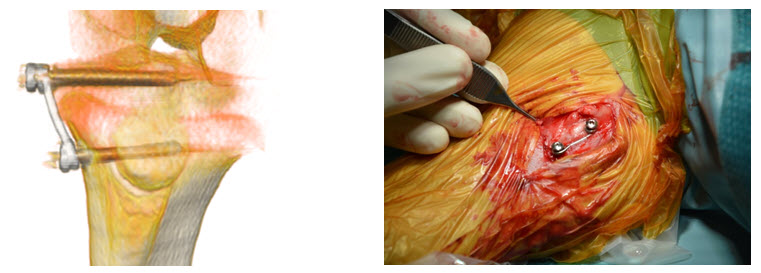
Thirty-six lambs were equipped with the proposed implant in order to create either varus-valgus deformities (phase 1, 18 lambs) or leg length discrepancies (phase 2, 18 lambs). They were assigned to 3 groups per phase considering 3 implant force levels (60 N, 120 N and 200 N). Changes in the medial proximal tibial angle (phase 1) and the developed differences in tibia length (phase 2) were assessed from biweekly radiographs.
The results show that the proposed implant concept enables safe and controlled growth regulation. Significantly different correction rates were achieved, depending on the applied force levels. Most efficient was the 200 N implant, followed by the 120 N one. Only marginal effect was observed for the 60 N implant. The correction rates remained constant over the treatment period, thus highlighting advantages with regard to more reliable treatment planning. No implant related failures were observed, underlining the passive concept safety. The concept revealed potential for clinical translation. Apart from the medical device development aspects, the results provide new important scientific information enhancing both the knowledge on bone growth processes and treatment of limb deformities.
-
Publication
Buschbaum J, Freitag L, Slongo TF, Zeiter S, Schütz M, Windolf M. Growth modulation of angular deformities with a novel constant force implant concept-preclinical results. J Child Orthop. 2021;epub April 7. https://doi.org/10.1302/1863-2548.15.200218
-
Partners
Slongo T (MD) Children’s University Hospital, Berne, Switzerland