AO Innovation Translation Center (AO ITC)
Empowering surgeons to improve patient care through innovation and evidence generation
In the AO ITC, our activities include:
-
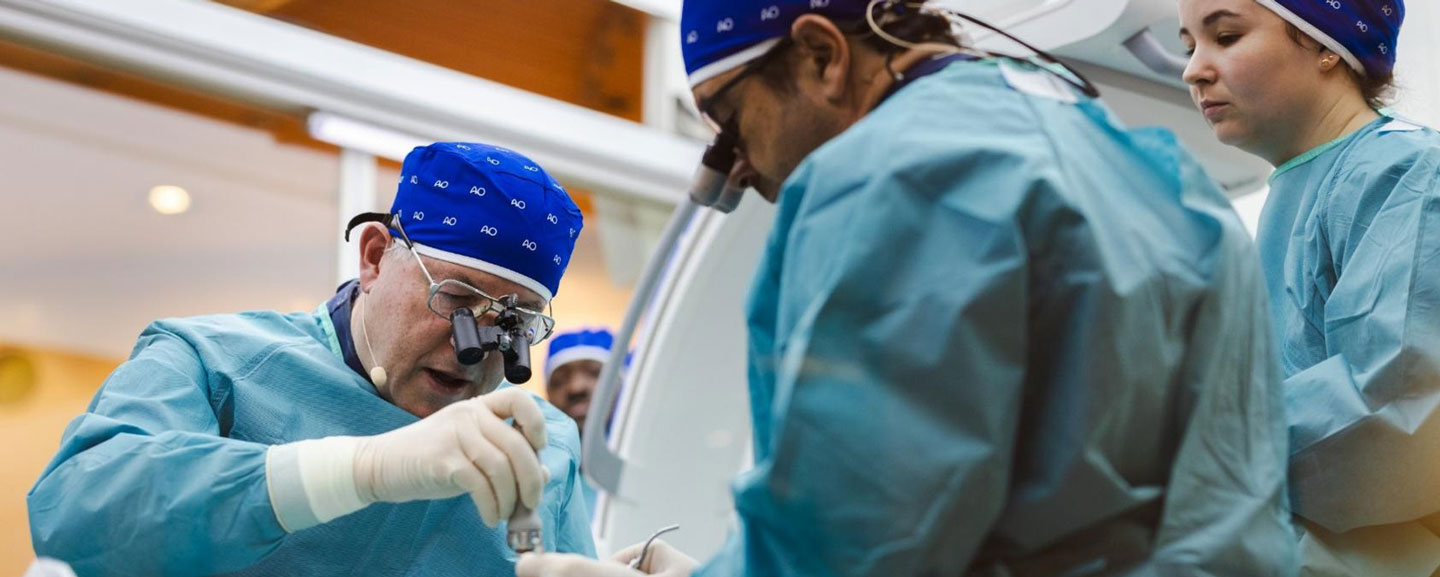
Gathering clinical needs: Via our global AO community of 400+ expert surgeons, we identify gaps in available solutions and define gold-standard treatments.
-
Collaborating with experts: We connect world-class surgeons with high-caliber partners to drive product innovation. Our portfolio numbers over 70 active development projects.
-
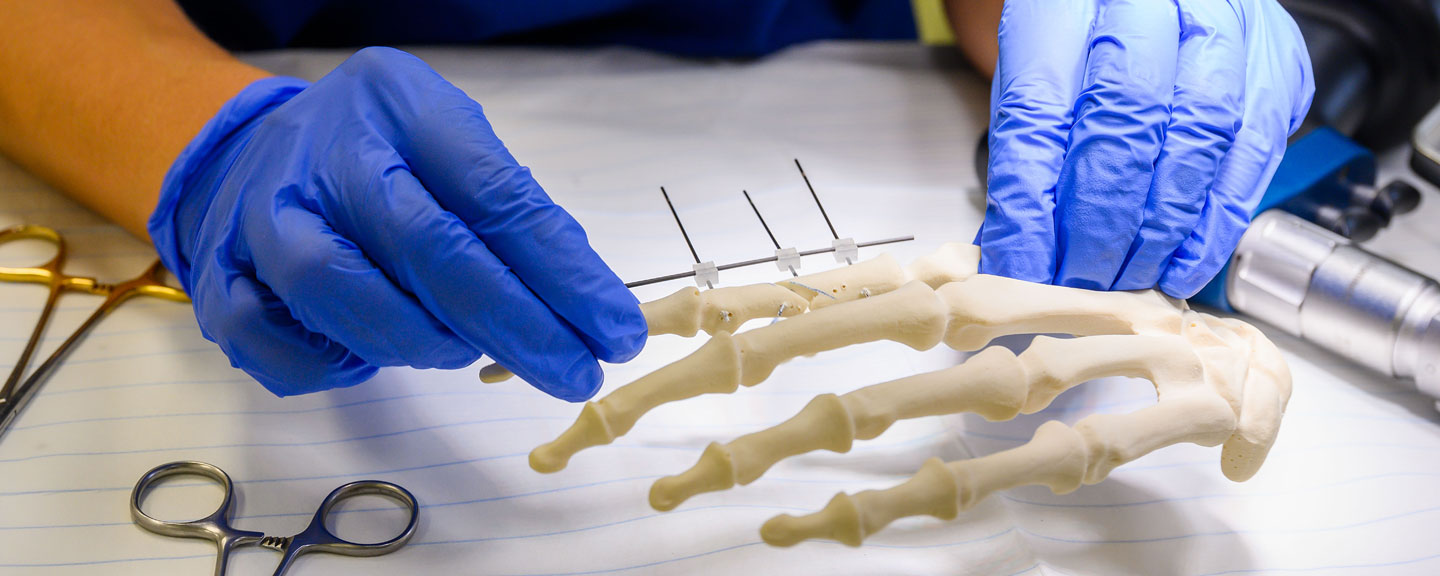
Delivering solutions: We develop novel surgical technologies through innovation and strategic investments.
-
Generating evidence: We deliver high-impact clinical research via 80+ ongoing clinical studies including over 10,000 enrolled patients. Our 750+ peer reviewed publications support better patient outcomes.
AO Technical Commission
Guiding the development and clinical testing of innovative surgical solutions

"Innovation at the AO has remained the same, finding solutions to clinical problems—meeting patient treatment needs. What has changed is implementation and the way that innovation is moving forward."
Robert McGuire, AO Past President
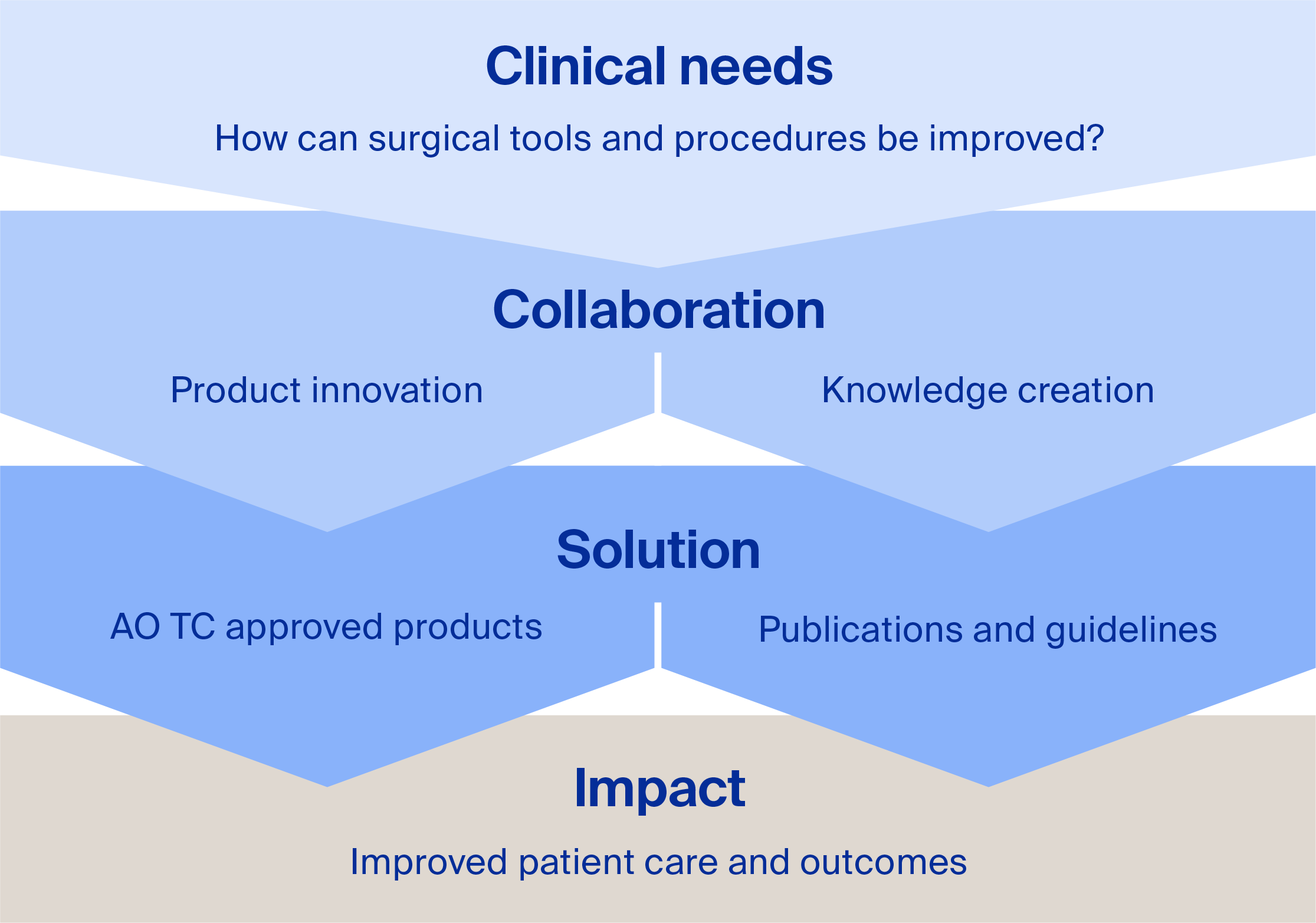
The AO ITC adds significant value to surgical innovations by identifying unmet clinical needs and driving collaboration in two key areas:
-
Product innovation: Our global network of expert surgeons collaborates with partners to develop pioneering surgical solutions.
-
Knowledge creation: We generate essential clinical evidence that supports optimal surgical treatments.
We deliver transformative solutions through AO TC approved products, publications, and guidelines. These advancements are disseminated via AO channels, ensuring the global surgeon community has early access to our innovative offerings. Our impact is ultimately to enhance patient care and outcomes.
Innovation news
-
Innovation
-
Innovation
-
Innovation