AO Small External Fixator
The AO Small External Fixator, developed toward proof of concept and valorization with AO development incubator resources made accessible by the AO Innovation Translation Center (AO ITC) Technology Transfer, was set for clinical documentation in summer 2021 in Germany, Austria, and Switzerland. The system which received Conformité Européene (CE) mark in early 2021 represents a soft tissue protecting, less bulky, and more efficient solution for treating bruised, fractured, or broken small bones compared with existing solutions.
With support from the AO ITC Technology Transfer, the AO Small External Fixator—invented by Austria-based surgeon Karl Heinz Bürger, MD—has been technically optimized regarding material choices, tolerances, and portfolio definitions, and received enthusiastic feedback from AO experts who tested it in AO Research Institute Davos (ARI) wet labs. With a first production run of 200 pieces completed, the solution is now ready for clinical application.
The clinical problem
Over the course of his career, one of the setbacks consistently experienced by Bürger was the instability of Kirschner wire (K-wire) fixation for fractures of the phalanx. Titanium plates—another fixation option—present additional challenges: "It's not always possible in an acute trauma situation to open the soft-tissue sufficiently to achieve fixation with a plate” says Bürger. Bone cement is also an alternative, but usage is expensive, time-consuming, and challenging because once it dries the fixation cannot immediately be corrected.
The clinical solution
Regularly faced with these constraints, Bürger set about to develop a solution: the AO Small External Fixator, which utilizes snap-on brackets to hold the horizontal rods of a fixation brace in place. With this concept in mind, he partnered with a product design expert who helped to create a prototype of a quick, affordable, stable, and tissue-preserving means of fixing fractures in small bones.
Development incubator resources made available through the AO ITC Technology Transfer have played a key role in the development of the AO Small External Fixator. Resources include investments granted by an independent board that approves proposals and consults extensively with experts in relevant fields. The board also offers knowledge about securing intellectual property, medical device development, enabling first clinical cases, and planning valorization of the proof of concept.
You might also be interested in
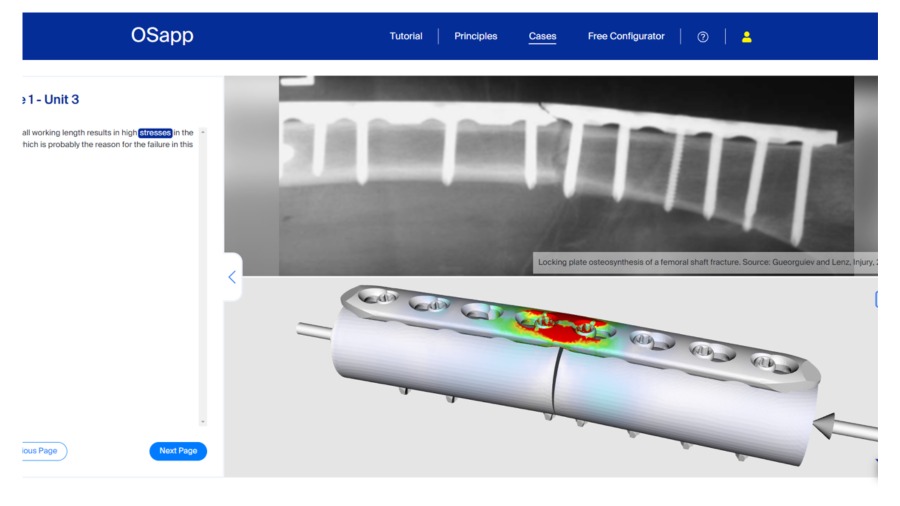
OSapp
A new, interactive, and constantly expanding virtual osteosynthesis software tool and interactive online learning platform.
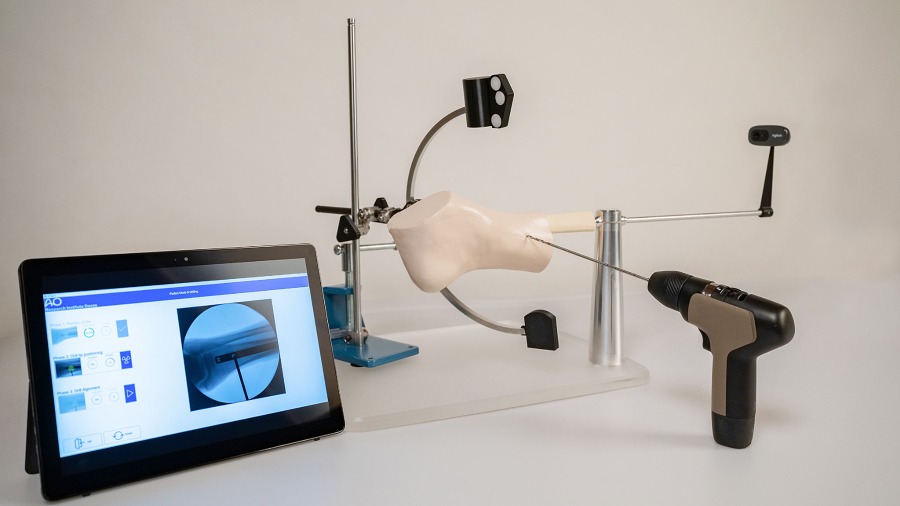
Digitally enhanced, hands-on surgical training
Digital innovation in surgical skill training.
Biphasic Plate
Improving fracture stabilization by providing a predictable mechanical environment at the fracture.
Technology Transfer of the AO fireside chat
AO Technology Transfer Board members Prof Anita Ignatius and Dr Robert Frigg say the AO is uniquely positioned to help innovators conquer those hurdles in order to improve patient care and outcomes.