AO CMF Start-up Grants
Calls for AO CMF research grant application open in Q4 every year.
AO CMF Start-up Grant Funded Research Projects
The abstracts of the latest funded projects under the start-up grant
-
AOCMF_S_2025-01323: Core Outcome Set for mandible reconstruction after mandibulectomy
Principal investigator: Saranda Ombashi, Erasmus MC, Netherlands
Clinical research
Abstract:
No structured and standardized manner to collect outcomes in patients with mandible reconstruction after mandibulectomy currently exists. Consequently, accurate data collection is impeded, leading to various problems with the assessment of treatment effectiveness across protocols and with effective benchmarking. In order to enhance progress and innovation in the field of mandible reconstruction, the establishment of a Core Outcome Set (COS) is essential.
Therefore, the aim of the proposed project is to establish a COS for mandible reconstruction after mandibulectomy , including the optimal type and timing of standardized outcome measures. Future implementation of this COS will facilitate data collection, benchmarking and other comparisons between patients, centers and countries.
Development of the COS will be done by our team from the Erasmus MC, that has extensive experience with the development, implementation and validation COS's in other fields of craniofacial surgery. The project will be supported with the expertise of AO ITC researchers, for which a grant proposal has already been approved by the AO CMF Research & Development Commission in Q4 2024. Similar to our previous COS projects, outcome measures for the COS will be selected throughout a modified Delphi study, including experts of the field and patients/patient representatives in the decision-making. Furthermore, attention will be paid to achieve a COS that represents both the patient's voice by the inclusion of valid and relevant Patient Reported Outcome Measures (PROMs), as well as clinical outcome measures.
By the end of two years, we expect to have a COS that is ready for implementation, meaning consensus is reached about the type of PROMs and clinical outcome measures and the exact moments of measurement. On the long term, we foresee this COS is implemented in daily practice and can be used internationally, similar to our previous COS initiatives in other craniofacial conditions. By combining the scientific expertise of AO ITC with our previous experiences in this field, we expect an optimal and efficient collaboration, making the goals of our project realistic within the expected terms.
-
AOCMF_S_2025-01520: Interrogating Fracture Microenvironment to Bioengineer Bone-on-a-Chip Device
Principal investigator: Jasmine Ho, Northwick Park Hospital, United Kingdom
Translational research
Abstract:
Facial fractures with malunion or non-union due to large critical mass defects present significant clinical challenges which often leads to suboptimal healing. Bone regenerative research has predominantly focused on long bone models, leaving a gap in understanding the specific healing mechanisms in facial bones, which are known to heal differently. This research aims to investigate the physiological microenvironment during facial fracture healing with an aim to develop a novel 3D bone-on-a-chip device which will be used to test biomimetic tissue-engineered (TE) products that promote bone regeneration.
The study will address significant gaps in understanding by characterising the dynamic microenvironmental conditions at the fracture site over time by utilising a rat model for mandibular fractures. This will allow for the development of a physiologically relevant and biological precise bioreactor microenvironment for bone-on-a-chips devices which will be used for the pre-clinical testing and optimisation of proposed therapeutic TE strategies, improving the clinical translatability of proposed TE therapies for mandibular defect repair. Using these devices will enable the necessary detailed exploration of the interplay between cellular activity, mechanical forces, and biochemical signals which will set the foundation for better understanding of orofacial bone repair and help develop targeted treatment strategies to enhance osteogenesis.
Potential TE solutions already in the pipeline within the lab are decellularised ECM hydrogel for bioprinting, the use of pre-conditioned mesenchymal stromal cells and cell-free exosomes, which will aim to accelerate bone regeneration and reduce complications associated with these facial fractures. Concomitantly, the development of bone-on-a-chip device will also support research sustainability by embracing the 3Rs—replacement, reduction, and refinement—by providing more accurate and ethical preclinical models which will reduce the reliance on in vivo models for future bone-related research. This project aligns with AO CMF's research mission by fostering translational research that bridges fundamental science and clinical application for bone defect repair.
-
AOCMF_S_2025-01517: Core Outcome Set for mandible reconstruction after mandibulectomy
Principal Investigator: Saranda Ombashi, Erasmus MC, Netherlands
Clinical research
Abstract:
No structured and standardized manner to collect outcomes in patients with mandibular reconstruction after mandibulectomy currently exists. Consequently, accurate data collection is impeded, leading to various issues with the assessment of treatment effectiveness across protocols and with effective benchmarking. In order to enhance progress and innovation in the field of mandibular reconstruction, the establishment of a Core Outcome Set (COS) is essential.
Therefore, the aim of the proposed project is to establish a COS for mandibular reconstruction after mandibulectomy, including the optimal type and timing of standardized outcome measures. Future implementation of this COS will facilitate data collection, benchmarking and other comparisons between patients, centers and countries. Ultimately, the identification of best practices will improve the care of the individual patient.
Development of the COS will be done by our team from the Erasmus MC, that has extensive experience with the development, implementation and validation COS's in other fields of craniofacial surgery. The project will be supported with the expertise of AO ITC researchers, for which a grant proposal has already been approved by the AO CMF Research & Development Commission in Q4 2024. Similar to our previous COS projects, outcome measures for the COS will be selected throughout a modified Delphi study. For this, experts on mandibular reconstruction from different regions and patients/patient representatives will be involved. Furthermore, attention will be paid to achieve a COS that represents both the patient's voice by the inclusion of valid and relevant Patient Reported Outcome Measures (PROMs), as well as clinical outcome measures.
By the end of two years, we expect to have a COS that is ready for implementation, meaning consensus is reached about the type of PROMs and clinical outcome measures and the exact moments of measurement. On the long term, we foresee this COS is implemented in daily practice and can be used multicentrically and internationally, similar to our previous COS initiatives in other craniofacial conditions. By combining the scientific expertise of AO ITC with our previous experiences in this field, we expect an optimal and efficient collaboration, making the goals of our project realistic within the expected terms.
-
AOCMF_S_2025-01518: Developing an Angiogenic Therapy to Enhance Repair of A Critical Size Jaw Defect
Principal Investigator: Jennifer Ten Eyck, University of Michigan, United States
Translational research
Abstract:
A critical size defect (CSD) in the mandible that is created by trauma will never heal on its own, causing patients immense, debilitating pain and severe difficulties with eating and speaking. The most current treatment options for CSD in the mandible include bone auto and allografts, free tissue transfers, distraction osteogenesis, synthetic bone substitutes as well as off label use of BMP. These reconstructive options suffer from associated donor site morbidity, multistage operative procedures, potential disease transmission, rejection, and black box warnings. (1-3) To date there are no existing therapeutics designed to utilize a vascular platform to augment the healing of a CSD. (3) This proposal aims to develop a therapeutic strategy designed to accelerate bone healing by generating increased vascular supply. Deferoxamine (DFO), an iron chelator, combined with Hyaluronic Acid (HA) has demonstrated a unique capacity to increase angiogenesis via the hypoxia inducible factor (HIF 1-alpha) pathway and substantially augment osteogenesis during bone healing.(3-7) Following promising results in long bone studies, our laboratory has since pioneered the use of this powerful strategy to accelerate bone repair and regeneration in the craniomaxillofacial skeleton. (8-12)
Aim 1: To gauge the extent of augmented angiogenic stimulation, vascular network formation, and bone regeneration within a murine mandibular CSD in response to HA-DFO implantation
Sprague-Dawley rats (n=30 per experimental group, total n=90) will undergo mandibular osteotomy and creation of a 5mm CSD with no treatment (negative control), one-time treatment of either HA-DFO (therapeutic group), or one-time treatment of recombinant human bone morphogenetic protein 2 (rhBMP-2) as a positive control at the time of surgery. The animals will be allowed to heal for 40 days prior to sacrifice and tissue analysis. We hypothesize that the negative controls will have severe limitations in healing that can be adequately quantified utilizing 3D microangiographic, μCT, and biomechanical data. Furthermore, we hypothesize that the HA-DFO will match or exceed the healing rate induced by rhBMP-2 as measured by microangiographic, μCT, and biomechanical metrics. As opposed to the adverse side effects associated with BMP, we expect that the healing of CSDs utilizing HA-DFO will occur without ectopic bone formation.
We believe that this novel and innovative new treatment will significantly change how CSDs and complex fractures are treated. Upon success, this award will provide the basis for subsequent large animal studies, further product development and eventually, human clinical trials. -
AOCMF_S_2025-01521: 3D-Printed Metal-Reinforced Biocomposite with Hydrogel for Jaw Reconstruction
Principal Investigator: Jane Pu, The University of Hong Kong, Hong Kong
Translational research
Abstract:
Background: Segmental mandibular defects require solutions balancing esthetics, mechanical strength, osteogenic potential, and infection resistance. Our group has developed a novel 3D-printed titanium (Ti)-embedded nano-hydroxyapatite (nHAp) biocomposite matrix, demonstrating excellent biocompatibility, mechanical strength and osteointegration potential for clinical application. However, direct implantation of the biocomposite risks infection and implant exposure in the long-term. The periosteum, as an important biological tissue, plays a crucial role in attachment, protection, support, and promotion of bone healing. Inspired by these functional properties, a gradient hydrogel coating on biocomposite is developed.
Aims and Objectives: This project aims to develop a hybrid scaffold with a sandwich structure, integrating a 3D-printed Ti-embedded nHAp biocomposite matrix as the core scaffold, a magnesium oxide (MgO)-embedded silk fibroin/tannic acid (SM/TA) bioadhesive as intermediate adhesive layer and an nHAp-embedded modified gelatin/hyaluronic acid (Gel/HA) based-hydrogel as the outer wrapping layer. This design aims to achieve optimal biocompatibility, gradient mechanical strength and osteoinductivity. Objectives include system design and fabrication to ensure effective integration between the layers; mechanical and biological assessments in vitro for design optimization, after which biointegration performance would be evaluated in rabbit mandible defect model in vivo .
Relevance: The hybrid scaffold addresses critical unmet needs in mandibular reconstruction by combining advanced materials and bone regenerative technologies.
Materials and Methods: The overall composite structure has a gradient mechanical strength decreasing from metal-reinforced biocomposite, to adhesive layer (SM/TA), then Gel/HA bioactive layer. In vitro experiments will assess the mechanical strength, biocompatibility, cell adherence, proliferation, and osteogenic potential. Preliminary In vivo study using rabbit mandible defect model will be used to assess implantation potential with bone regeneration and biointegration.
Expected Results: The hybrid system is expected to demonstrate a decreasing gradient mechanical strength from biocomposite, adhesive layer to bioactive layer, while maintaining excellent osteogenic and vascularization properties. The hydrogel outer layer is anticipated to enhance biointegration and reduce long-term implant exposure risks. Long-Term Goal: The overall objective is to develop a clinically viable, hybrid reconstructive solution for segmental mandibular defects that combines mechanical robustness and osteogenic potential.
-
AOCMF_S_2025-01522: Ischaemia and reperfusion injury (IRI) in free microvascular human transplants
Principal Investigator: Jonas Wüster, Universitätsklinikum Freiburg, Germany
Clinical research
Abstract:
This explorative prospective study aims to evaluate ischemia-reperfusion injury (IRI) in free microvascular transplants by analyzing various flap types, including the radial forearm free flap (RFF), free fibula flap (FFF), anterolateral thigh (ALT) flap, deep circumflex iliac artery bone (DCIA) flap, free scapula flap (SFF), and latissimus dorsi flap (LDF). The study will investigate IRI across different tissue compositions—fasciocutaneous, myofasciocutaneous, and osteomyocutaneous—by collecting tissue and blood samples at standardized time points: before ischemia, during ischemia, and after reperfusion. Histological and stimulated raman histology will be conducted to characterize the cellular and molecular changes associated with IRI. In addition to tissue sampling, venous blood will be drawn directly from the vascular pedicle to assess systemic responses. Clinical parameters will be systematically recorded, and patient outcomes will be monitored to correlate biological findings with surgical success. By gaining new insights into the IRI in autologous microvascular transplants, an improved surgical outcome can be achieved by identifying potential negative and/or positive influencing factors.
-
AOCMF_S_2025-01523: Involvement of Osteal Macrophages on Bone Formation in Inflammatory Conditions
Principal Investigator: Reon Morioka, Shimane University Faculty of Medicine, Japan
Translational research
Abstract:
Background: Osteal macrophages (OsteoMacs) play a critical role in regulating inflammatory responses and tissue regeneration during bone formation. In general, bone formation is promoted in environments dominated by M2 macrophages (M2Φ), which enhance tissue regeneration. Interestingly, however, in oral and maxillofacial surgery, new bone formation is often accelerated even under inflammatory conditions, such as mandibular osteomyelitis.
Aim: This research aims to investigate the mechanisms underlying periosteal reactions in inflammatory conditions by clarifying the functions of OsteoMacs surrounding the periosteum in inflammatory jawbone diseases. It is hypothesized that the interaction between periosteal cells (PCs) and OsteoMacs contribute to new bone formation even in the presence of inflammation.
Materials and Methods: Human bone specimens will be collected during surgery from four groups:
- control intact bone
- chronic osteomyelitis
- medication-related osteonecrosis of the jaw (MRONJ)
- osteoradionecrosis (ORN).
The 3D surgical guide sampling technique will enable precise correlation between bone architecture and histological features. Analyses will include micro-CT for bone quality, immunohistological evaluation of bone and periosteum, and gene expression profiling of periosteal tissues. The polarization properties of OsteoMacs will be assessed, and PCs from each group will be characterized using single-cell RNA sequencing (scRNA-seq). Their osteogenic potential and influence on THP-1-derived macrophage polarization will be evaluated through in vitro co-culture systems.
Expected results: The interaction between PCs and OsteoMacs will be a critical determinant of bone regenerative capacity. By characterizing and matching the profiles of periosteum and OsteoMacs across various inflammatory jawbone diseases, we aim to clarify the mechanisms that drive bone formation under inflammatory stress. Furthermore, the use of surgical guide techniques interestingly offers an innovative approach by enabling precise alignment of CT, micro-CT, and histological imaging with in vitro phenotyping of the periosteum.
Overall objective: The results in the present research can be beneficial for future strategies in bone regenerative medicine, including the development of cultured periosteum sheet implants capable of controlling OsteoMac polarization to enhance bone regeneration. This approach could address clinical challenges of bone regeneration procedures in both oral and maxillofacial and orthopedic surgery, particularly in inflammatory environments.
-
AO CMF_S2024_00012: Mandibular reconstruction utilizing advanced patient-specific implants (Netherlands)
Investigators: Brend Pjotr Jonker, DDS, MD, PhD, Nasim Shahriar M.Sc. Abdullah Tahir Şensoy PhD, Mohammad J. Mirzaali PhD Amir A. Zadpoor PhD Eppo B. Wolvius DDS, MD, PhD
Abstract of full proposal:
Mandibular reconstruction is one of the more complex surgical operations that cranio-maxillofacial surgeons face. It has been reported that in 2020, there were around 377,000 new cases of oral cancer globally, frequently requiring resection of the mandibular bone followed by reconstructive surgery. Additionally, significant reconstruction of function is necessary for benign lesions, osteomyelitis, osteoradionecrosis, and extensive trauma.
Common mandibular reconstruction surgery techniques include free vascularized osteocutaneous flaps and fixated with regular, pre-bend or patient-specific plates. These procedures are invasive operations that often lead to considerable donor site morbidity, chronic pain, infection, and does not always yield optimal functional or aesthetic results. Additionally, the use of conventional plates is often associated with complications such as screw loosening, plate exposure, and plate fracture. Even though the implantable solutions such as patient-specific plates, cages and scaffolds have been explored and shown comparatively better results in improving surgical techniques in mandibular reconstruction, they have also common drawbacks, such as screw loosening, implant or bone failure due to fatigue and stress shielding effect, and poor bone remodeling at the implant site.
Meta-biomaterials with their distinct mechanical and biological properties, are a promising method to address all the above-mentioned issues in designing multifunctional implants. The unit cell type, pore size, porosity percentage, and the distribution order of the defined unit cells throughout the implant can tune the mechanical and biological properties of the implants. This proposal, therefore, proposes the state-of the-art hybrid design of mandibular implants using meta-biomaterials, considering the physiological loading condition of the implant to develop an implant that can withstand the loads applied while promoting cell attachment and bone growth without the need for free vascularized osteocutaneous flaps.
In this regard, computational modeling and adaptive bone simulations will be developed to evaluate the effect of various distributions of auxetic and non-auxetic unit cells throughout the implant to optimize their strain distribution, fatigue behavior and osteoconductivity at the mandibular site in the scenario of a segmental defect obtained by surgical resection. Starting from an existing workflow to design patient specific mandibular implants, we propose to create phenomenological prediction models to evaluate bone remodeling process surrounding the implant. The two simulations would then be combined to create a computational model including both mechanical and biological behaviors surrounding a hybrid auxetic non-auxetic mandibular implant. We hypothesize that hybrid implant design can overcome the current issues with the conventional mandibular cages.
-
AO CMF_S2024_00281: Fabricating Pre-Vascularized Anatomical Grafts for Craniofacial Rehabilitation (Israel)
Investigators: Idan Redenski / Samer Srouji, Galilee Medical Center, Israel
Abstract:
While in the past 25 years, head and neck cancer has shown a decline due to prevention and early diagnosis, the incidence of oral cancer has been on a rapid rise, especially among young individuals without any known risk factors. Oral cancer and oral squamous carcinoma (OSCC) inflict more than 300,000 new cases and 145,000 deaths annually, with the aggressive nature of SCC necessitating aggressive resections of soft tissues with supporting bony framework with wide tumor-free margins. Spontaneous healing of substantial oro-facial defects, together with compromised vascular support, is unrealistic. Specifically, the adverse microenvironment created at the defect site may hinder the innate regenerative capability of the body, thus forcing external intervention by surgeons to try to repair the damaged tissues. These procedures necessitate the harvest of large, vascularized bone-containing autologous grafts.
The current state-of-the-art techniques utilize vascularized soft and hard tissue flaps, which have improved orofacial reconstruction outcomes in recent decades. However, success rates are still limited by the extent of bone tissue vascularity and lack of oral soft tissues. Autologous grafts are still associated with significant disadvantages, such as limited volume, tissue site morbidity, and substantial post-operative complications. Titanium alloys, despite excellent biocompatibility and high mechanical strength, are not biodegradable, cannot replace the missing bulk volume of bone tissue, and remain as permanent implants within the body. Moreover, these biomaterials can only bond with native bone tissue through mechanical interlocking with no bioactivity, which can result in loosening and wear, adversely affecting reconstructed soft tissues. Thus, maxillofacial surgeons and tissue engineers are confronted with the same substantial challenges in the field – creating biologically inspired, human-sized tissue replacements while minimizing the harvest of patients' tissues.
Recently, 3D-printed bone scaffolds have been experimented with to reconstruct significant defects, and different material deposition strategies have been able to recapitulate complex anatomies. Moreover, human-scaled mandibular bone was fabricated by using a tissue-engineered approach based on a biomimetic ECM-based bone matrix, harnessing the innate regenerative potential of the host's body. Still, Survival of tissue grafts at a surgical site critically depends on host capillary invasion into the tissue constructs. Laboratory-made 3D-printed engineered grafts are still discrete elements, without an arteriovenous blood supply and lacking constant connectivity with the host circulation. Despite the progress in both tissue engineering approaches and the development of new biomaterials, the lack of initial post-implantation vascularization and host-to-graft vascular connectivity is a primary cause of failure in the engraftment of bioengineered constructs.
Herein, we propose to employ accurate 3D printing to fabricate soft and hard tissue constructs, whereby a printed provisional mold will be used to manufacture engineered grafts for craniofacial rehabilitation. First, 3D printed constructs will be populated with a biomimetic hydrogel loaded with human mesenchymal and endothelial cells, which will support both vascularization and osteogenesis in vitro. Engineered grafts will be further vascularized by inducing axial vascularization, guided by a predefined architecture within constructs. Finally, the composite grafts, now in the form of soft and hard neo-tissues, will be used to rehabilitate soft and hard tissue defects in an in-vivo murine model. Achievement of this translational proof-of-concept study holds the potential to provide a solution for a clinical unmet need to reduce morbidity and improve recovery of SCC patients following resection operations.
-
AOCMFS-24-07S Project title: Automated segmentation of bone invasion in oral squamous cell Carcinoma
Applicants: Steybe D / Obermeier K / Poxleitner P / Russe M F / Sabel B / Otto S
Abstract:
Recently, Artificial Intelligence (AI) applications have revolutionized a variety of medical fields, including medical image segmentation. With new Convolutional Neural Network approaches, precise automated segmentation of the complex anatomy of the head and neck has become possible; this does include bone structures (mandible, viscerocranium, skull base), foramina/canals, paranasal sinuses and soft tissue (ocular globe, optic nerve, extraocular muscles).
Given 377,713 new cases of oral squamous cell carcinoma (OSCC) reported in the 2020 Global Cancer Statistics and the fact that bone invasion is found in up to 56% of OSCC cases [2], automated segmentation of bone invasion in OSCC could make a valuable contribution in this field. Bone invasion and especially medullary bone invasion is of high relevance for the determination of the appropriate staging and treatment strategies for OSCC [3], and an automated approach could reduce the human resources required and speed up identification of this therapeutically and prognostically relevant parameter. From a surgical perspective, automated segmentation approaches will generate the high-quality 3D models required for accurate CAD/CAM-based resection and reconstruction of the bone, significantly accelerating this procedure and reducing the human resources required for personalized cancer surgery.
The proposed project will implement an open source-based platform (NORA Imaging, Department of Diagnostic and Interventional Radiology, University Medical Center Freiburg, Germany) for remote automated segmentation of radiographic imaging data from the head and neck region. This will allow us to include > 100 CT scans of OSCC patients with bone invasion from two major Oral and Maxillofacial Surgery centers (University Hospital LMU Munich and University Hospital Freiburg) and thus generate the data required to evaluate the applicability of automated segmentation of bone invasion in OSCC.
Validation of the results will be performed using established evaluation metrics; moreover, clinical and pathohistological evaluation of the results will be performed. -
AOCMFS-24-04M Project title: Innovative method of angiogenesis morphometric analysis in large preclinical models of jaw-bone regeneration
Applicants: Mangione F / Vital S / Salmon B / Bardet C / Jacobs R
Abstract:
INTRODUCTION:
In the oro-facial region, tissue engineering is required to recreate an adequate bone volume for implantology rehabilitation purposes either to treat bone defects resulting from traumas, malformations, or neoplasia.The assessment of angiogenesis is of primary importance for jawbone tissue engineering since blood vessel formation is crucial for bone regeneration.
In preclinical large animal models of jawbone regeneration, non-destructive and non-invasive methods for the evaluation of tissue regeneration are required, according to 3Rs principles (Replacement, Reduction and Refinement). Cone beam computed tomography (CBCT) angiography can be interestingly implied for characterization of angiogenesis during bone regeneration.
The aim of this study is to validate an innovative method of blood supply morphometric analysis using an adapted CBCT subtraction angiography technique in a porcine model of jawbone regeneration.
MATERIALS AND METHODS:
This study is designed in conformity to the ARRIVE guidelines and approved by the Ethics Committee for animal experiments of French Ministry of Research (APAFIS#12534-2017121118586862). 2 eleven-months-old Yucatan minipigs will be used. A buccal wall bone defect will be surgically created in the premolar-molar area in each hemimandible. Angiogenic process will be monitored during 90 days by CBCT angiographies. Images will be acquired by C-arm CBCT.Scanned volumes will be imported into MeVisLab (MeVis Research). Vascular net of the region of the defect will be segmented after the subtraction of mineralized tissues.
Segmented blood supply will be quali-quantified by morphometric analysis. Considered parameters will be: number of edges, number of nodes, radius of the edges, length of the edges and number of edges per node.Histology and immunohistochemistry highlighting angiogenesis (CD31, a-SMA) will be performed and blood vessels will be quali-quantified. Data from histology and imaging morphometric analysis will be statistically compared by Bland-Altman analysis.
CLINICAL RELEVANCE:
CBCT subtraction angiography for angiogenesis assessment in large preclinical models could substantially contribute to the validation of jawbone regeneration procedures. Since the technique allows to perform longitudinal non-destructive follow-up of vascularization, it could be interestingly employed in other pre-clinical studies of dento-maxillo-facial tissue engineering procedures as well as in pathological models such as BRONJ ones. -
AOCMFS-23-18M Project title: Developing multi-functional patient-specific mandibular implant using micro-additive manufacturing
Applicants: Mirzaali M / Shahriari N / Jonker B
Abstract:
The mandible is the largest bone in the human skull and acts an important role in the body, however, its health and functionality can be affected by diseases such as bone infections, neoplasms, and trauma. These defects can severely cause facial deformity, loss of speech, and reduced masticatory function if left untreated. To restore the functional and esthetical role of the mandible, it is essential to perform reconstructive surgery. The currently involved techniques for mandibular reconstruction comprise the use of traditional vascularized combined soft and bony tissue flaps which encompass several drawbacks such as the limitation in the amount of available bone, increased risk for surgical revisions, and prolonged recovery processes. Therefore, it reveals the importance of developing mandibular implants with (bio)functional properties as well as adjusted shape within which those advanced mandibular implants can provide a faster tissue integration without increasing the risk of implant-associated infections.Here, we propose an innovative design and manufacturing process for the development of multi-functional patient-specific mandibular implants. In order to promote better cell-implant integration, we propose surface-based lattice patterns in the functionally graded porous structure of the mandibular implants. This will allow us to not only mimic the mechanical and morphological properties of the natural bone but also to increase the contact area between the implant and the remaining bone, thus improving the integration of the implant with the undamaged tissue. In addition, we will develop a localized and spatiotemporal functionalized surface coating using a combination of advance manufacturing processes (i.e., additive manufacturing, AM) and atomic layer deposition (ALD) techniques. Such a combination will enable us to coat the surface of the additively manufactured porous implant and provide on-demand bio-functionality (i.e., revealing anti-bacterial properties over time).
In this project first, the innovative design workflow will be developed. Then, a selective laser melting (SLM) machine will be used to manufacture the developed design with different groups of Ti-6Al-4V powders and antibacterial materials will be deposited on their surface by ALD technique. The outputs of this proposal will go beyond the state-of-the-art and will propose next-generation mandibular implants with multiple bio-functionality necessary for craniomaxillofacial surgeries. -
AOCMFS-23-15H Project title: Modification of the pull-through approach using a v-shaped osteotomy in posterior oral and oropharyngeal carcinomas. A pilot study
Applicants: Hofmann E / Neckel N / Kreutzer K / Koerdt S
Abstract:
The project is designed as a pilot study to demonstrate the clinical feasibility of a modified pull-through approach for the resection of head and neck malignancies, which are localized at the posterior third of the oral cavity and the oropharynx. Current surgical techniques, such as the mandibular lip-split approach and the pull through approach with a lingual release, are associated with morbidities, including difficulties in swallowing and speech. The motivation of this study was to introduce a modified pull-through approach to reduce postoperative morbidities and improve patient’s quality of life.
The modified pull-through technique uses individualized CAD/CAM-designed cutting guides to facilitate a v-shaped, self-retentive osteotomy to maintain the attachment of the geniohyoid und genioglossus muscles. By maintaining the physiological attachment of these muscles, that are considered essential for swallowing and speech, postoperative function will be improved. The v-shaped osteotomy eliminates the need for osteosynthesis to re-fixate the bony segment in contrast to the mandible lip-split approach. Our research group was able to show the feasibility of the modified pull-through technique and the anatomic foundation for its clinical use in a cadaver study.The aim of this pilot study is to demonstrate the clinical application of the described modified pull-through approach. We hypothesize that the improved postoperative function with regard to speech and swallowing benefits patients and improves their quality of life. By eliminating the need for osteosynthesis material to restore bone continuity, postoperative complications can be reduced.
-
AOCMFS-23-11A Project title: A multiscale, biology-based strategic approach to optimizing cleft repair outcomes
Applicants: Aello F / Koudstaal M / Helms J
Abstract:
In patients born with cleft lip and palate (CL/P), surgical repairs are typically undertaken in the first years of life. This approach is guided by clinical data demonstrating that early closure of the lip and soft palate improves feeding and speech development 1,2. There is, however, no consensus on the optimal timing for closure of the hard palate because, in this anatomical location, early surgical repair is associated with a subsequent arrest in midfacial growth 3-7. This growth arrest is directly attributable to the surgical intervention since children who undergo similar repairs after their fifth birthday rarely exhibit midfacial growth truncations 3-7. If we understood exactly what aspects of the surgery led to craniomaxillofacial (CMF) growth arrest in younger patients, then we would be in a better position to develop new techniques and strategies that significantly improved overall functional outcomes 8.Currently, there is a limited understanding of why hard palate repair procedures cause surgically-induced midfacial growth arrest. It has been speculated that general disruptions at the vomero-premaxillary and transverse palatal sutures, scarring in soft tissues, and/or resorption/remodeling of hard tissues are contributing factors 9-11 but at present, these data are insufficient to guide surgical improvements. The purpose of our proposal is to use a validated animal model, coupled with a surgical intervention that mimics hard palate repair and reliably produces midfacial growth arrest, to pinpoint those soft and hard tissue responses that specifically contribute to midfacial growth arrest. With that information in hand, we then propose to test whether modified surgical approaches can minimize growth disturbances and yield better overall outcomes for CLP patients.
-
AOCMFS-23-06M Project title: Automated image to patient registration in augmented reality navigation using deep learning models
Applicants: Benmahdjoub M / Wolvius E / Niessen W / Walsum van T
Abstract:
In the past decades, surgical navigation has shown to be of important assistance to surgeons. This technology allows the localization of instruments with respect to a preoperatively acquired image (e.g., CT, MRI). It also helps with visualizing preoperative planning to avoid critical structures or to reach a specific target during the operation. A crucial step to benefit from these advantages is image to patient registration. This procedure aligns the preoperative image with the real patient. Conventionally, it is a manual process where the surgeon uses a pointer to locate landmarks (anatomical/artificial) on the patient, that have also been annotated on the diagnostic preoperative image. The two sets of points, preoperatively annotated and intraoperatively pointed, are used to compute the alignment matrix [1]. Whereas more advanced approaches, such as scanning the patient skin surface, have been developed, the alignment remains a relatively elaborate part of using navigation. In addition, the use of conventional navigation systems (tracker and 2D screen), is hampered by the hand coordination difficulty, the lack of depth perception, and the switch of focus between the surgical area and the screens [2].Augmented reality has been suggested in the literature as a potential solution to these issues and has been linked to many craniomaxillofacial surgeries [3].In augmented reality (AR) navigation, the AR device sensors may aid in automatic detection of landmarks, and thus it may also help in a more automated alignment of the patient to image in AR-based navigation.
The automated detection of artificial markers (such as sticked to patient) is an important step in facilitating this alignment, and if the AR system is linked to a conventional navigation system, it enables automation of the image to patient registration procedure. Such automated approaches may also permit continuous quality check of the patient-to-image alignment in navigation. Whereas deep learning-based methods likely are a suitable approach to such automated detection of markers, the training of models is hampered by the lack of real data (pictures of patients). It is our hypothesis that such models may also be trainable by synthetic images, generated from photo-realistic rendering engines. Demonstrating that this indeed is possible opens the way for novel AI approaches in surgery, where real patient data for training is lacking.
Therefore, in a first step, we will generate photo-realistic synthetic data, and investigate its use for landmark detection on real images. In a second step, we want to investigate the usability of the trained models for image to patient registration in an augmented reality navigation context.
-
AOCMFS-22-23K Project title: MOBILIZING MECHANOBIOLOGY OF PERIOSTEAL CELLS FOR CRANIO-FACIAL BONE REGENERATION
Bone periosteum is a natural scaffold and a source of regenerative cells and bioactive factors that promote osteogenesis. The areas in which the periosteal cells are subjected to mechanical loading co-localize with the greatest new bone deposition. The regenerative pathways during periosteal distraction osteogenesis (PDO) depend on the duration and magnitude of tensile stress. Our preliminary data indicate that the distraction alternated with relaxation, named “pumping of periosteum” (PP) results in enhanced osteogenic capacity of periosteum. While the histological features associated with PDO have been described, the underlying molecular mechanisms remain unknown. Our objective is to clarify the interaction between the periosteum and the underlying tissues in response to mechanical stimuli, by using a rat model of calvarial regeneration. In parallel, the same studies will be conducted using a mechanical loading bioreactor, a model that allows extension to controlled studies of human bone regeneration. To this end, we intend to: 1. Examine bone formation following PP protocol at different time points (Aim 1), and 2. Analyse the expression of bone-specific genes on the mRNA and protein levels (Aim 2). Following a latency period, the PP protocol alternating relaxation with activation will be systematically compared to the conventional PDO protocol. The amounts of (i) new tissue generated relative to the area confined by the parental bone, (ii) periosteum and (iii) distraction plate, will be determined histomorphometrically and by micro-CT. In both models, molecular differences between the two modes of periosteal elevation will be related to the alterations in the expression of tissue-specific proteins. We expect that the parameters of periosteal manipulation will induce expression of osteogenic differentiation markers and influence the nature and kinetics of bone formation in places that would otherwise never become bone. The osteogenesis and the subsequent early stage bone development shall involve a mechanism, which detects and responds to the level and duration of hydrodynamic shear forces. Consequently, we propose to elucidate the mechanobiological principles governing hard tissue formation during different modes of periosteal elevation. The outcomes of this work would impact our understanding of the role of periosteum in bone augmentation and ultimately, enable a paradigm shift in the field of regenerative medicine.
-
AOCMFS-22-18K Project title: Preliminary clinical study on the treatment of segmental mandibular defects using a Ti-cage implant
The current standard procedure to restore mandibular continuity defects involves free tissue transfer with an autologous composite bone flap. Even though success rates are high, several critical drawbacks have been associated with this procedure, including severe donor site morbidity, long hospital stay and recovery process, need for high surgical expertise, insufficient bone graft height, and mechanical failure of the plating system. A systematic approach for the designing and testing of patient-specific implants used as an alternative to the free flap procedure appears to be still lacking.
The goal of this project is to conduct a preliminary clinical study on the reconstruction of a lateral segmental defect following mandibulectomy using a 3D printed patient-specific titanium cage implant. Qualified patients will require reconstruction following segmental resection for a benign defect in the lateral mandible and have no indication for radiotherapy. In these cases, a normal viable and adequately vascularized soft tissue bed is expected. The reconstruction cage will be designed through a semi¬-automatic workflow for designing patient¬ specific mandibular reconstruction implants in the Materialise Mimics® and Materialise 3-Matic® software environment. Comparison of specific implant design variables (e.g. number of screws, screw position, and screw type) will be made prior to clinical application using topology optimization techniques. The biomechanical performance of the different implant designs will be evaluated through validated computational finite element analyses and experimental testing. The porous and cage-shaped design of the implant provides the possibility for the insertion of autografts harvested with minimal donor side morbidity, as well as the subsequent integration of dental implants. Post-operative monitoring using (CB)CT scan imaging following surgery will take place to quantify bone ingrowth and graft healing. Aesthetical and functional outcomes of the reconstructive procedure will be assessed using a patient-perceived quality of life questionnaire. A successful in-house study on the semi-automated implant designing, computational modeling and biomechanical testing of a patient-specific proof-of-concept implant highlights the potential of the proposed framework to yield more cost- and time-effective pre-surgical planning and result in implant designs that can minimize morbidity and maximize aesthetic and functional outcomes.
-
AOCMFS-22-09G Project title: Fractional laser combined with self-regenerating cartilage to repair the temporomandibular jointAfter chronic low back pain, temporomandibular disorders (TMD’s) are the second most common musculoskeletal condition affecting 5-12% of the population, with an annual health cost burden estimated at $4 billion worldwide. Temporomandibular joint osteoarthritis (TMJOA) is a subtype of TMD characterized by slow and progressive degeneration of the mandibular condyle cartilage and bone, dramatically impacting function and quality of life. The aim of this study is to use a novel approach that evaluates the use of fractional laser treatment in cartilage combined with our method for generating new cartilage matrix using dynamic Self-Regenerating Cartilage (dSRC) to regenerate articulating cartilage defects in a rabbit TMJ model. To form the dSRC, freshly harvested rabbit ear chondrocytes will be placed into sealed 15-mL polypropylene tubes and cultured on a rocker at 40 cycles per minute for 14 days at 37°C. Chondrocytes aggregate and generate new extracellular matrix forming a pellet of dSRC. Microchannels of approximately 300-500 µm diameter will be created by infrared laser ablation in freshly harvested rabbit TMJ condyles. To evaluate cartilage generation ex vivo, the dSRC aggregates will be implanted in laser-ablated microchannels in rabbit TMJ condyle articular cartilage. dSRC samples after 2 weeks of in vitro culture and samples of native articular cartilage will be stained with H&E to evaluate chondrocyte density and neocartilage integration. Safranin O (sulfated GAG) staining, and Toluidine blue (proteoglycan) staining will also be performed to assess the biochemical composition of the neomatrix. To evaluate this combined approach in vivo, 16 New Zealand white rabbits will be used. Both TMJ condyles will be surgically accessed using a minimally invasive approach developed by our group. Laser-ablated microchannels will be created in the TMJ condyle cartilage and two groups will be studied: Group I – laser-ablated microchannels (empty; n=16 condyles) and Group II – laser-ablated microchannels+dSCR (n=16 condyles). Rabbits will be euthanized at 4 (n=8) and 8 (n=8) weeks, and the TMJ condyles will be harvested for processing. Condyle articular cartilage regeneration will be performed using micro-CT analysis, histological analysis (H&E, Masson’s Trichrome, Safranin O, and Toluidine Blue staining to assess the biochemical composition of the neomatrix) and immunohistochemical analysis of collagens type I (fibrocartilage) and type II (hyaline cartilage).
-
AOCMFS-22-05C Project title: Augmented reality in Head and Neck surgery. Use of the Hololens2 in planning and performing a fibular free flap reconstruction for a facial hard-tissue defect
Background:
Augmented Reality (AR) is the projection of a computer created image onto the observer’s real environment. AR can allow the projection of medical images such as CT scans onto a patient during surgery. This additional information can help guide surgeons during the procedure, showing the surgeon where to place bone cuts, screws and prostheses. This will allow the surgeon to see patient’s anatomy beyond the surgical incision. Therefore surgery can be performed through a smaller cut. Currently, we use cutting guides on bones which act like stencils. In contrast to AR guides, cutting guides are physical objects. They take weeks to produce and are not re-usable.
Aim:
To compare the accuracy of AR guided bone cuts and cutting-guide guided bone cuts against pre-operative planning.
Method:
30 surgeons in total will be recruited and asked to produce a total of 18 cuts on 3D printed fibula models using a surgical saw and fissure burs. 6 cuts will be made using AR guidance, 6 using cutting guides (control group) and 6 using AR guidance to locate the cutting guide onto the model. The produced bone cuts will undergo CT scan and analysed digitally.
Results:
Dice-coefficient and Hausdorff analysis will be used to compare the cut bone models and the pre-operative planned models. This will allow comparison of the 3 experiment arms.
Conclusion:
If AR is shown to be within acceptable accuracy to guide bone cuts, this experimental model can be used in cadaver studies. AR guided surgery has the benefit of having a short turn-around, it can be modified intra-operatively by the surgeon. It is a much cheaper system and better environmental sustainability profile. This is because the AR Head Mounted Device is reusable.
The same technique can be subsequently applied to other areas of CMF surgery. In CMF trauma, the facial bones are often broken into many small pieces. AR projection of CT scan will allow all the bone fragments to be seen virtually without being fully exposed surgically. It will allow on-table visualisation of hard tissue, minimising surgical access and morbidity.
-
AOCMFS-21-31V Project title: Towards the application of augmented navigation with HoloLens in orthognathic surgery
Computer-aided technologies have revolutionized orthognathic surgery (OS) facilitating cephalometric analysis, surgical simulation, and the fabrication of splints and patient-specific implants. Despite the increasing body of evidence that these techniques are improving accuracy of surgical outcome, wide implementation is limited due to relatively high costs and the time-consuming and complex handling of the software. Alternatively, the application of navigation systems for OS could potentially speed up part of the process through bypassing the necessity of intraoperative surgical guides and splints. So far, navigation has been successfully used for various CMF surgical interventions such as mandibular reconstruction, bony contouring, and maxillary repositioning (2). However, the use of conventional navigation systems for surgery faces three challenges: the depth perception, the hand-eye coordination, and the continuous switch of focus between the navigation screen and the operation site.
Augmented reality (AR) helps in addressing these challenges. This technology brings 3D patient-specific data into the view of the surgeon. By using an AR device, drilling paths, and cutting planes are visualized at the right location, thus eliminating the need to look away from the surgical site. Additionally, AR headsets provide a stereoscopic view of the 3D model which makes it easy to understand the 3D shape of the targeted anatomy. We developed a new augmented reality navigation system with an integrated HoloLens 2 for surgical purposes, that allows an integration of an AR device with a conventional navigation system . The method does not require any adaption of the AR device, and uses a multi-modal marker for aligning the AR device with the navigation system’s coordinate system. The use of this system requires a thorough pre-clinical evaluation. Therefore, four surgeons will use the AR navigation system in a series of phantom trials. The phantoms consist of 3D printed mid-face regions of 20 patients where the drilling trajectories and the cutting planes on each phantom is planned preoperatively. Subsequently, the planning data is imported into the AR system using the HoloLens 2 which projects the 3D virtual models on top of the phantom models. The operators will perform the procedure assisted by the AR visualization, and the comparison between postoperative outcome and preoperative planning will determine the system’s performance.
-
AOCMFS-21-23A Project title: microRNAs: new tools for craniomaxillofacial bone repair (miR4CMF)
microRNA (miRNA) therapeutics have emerged as a promising strategy for a broad range of diseases, yet their use for craniomaxillofacial applications remains largely unexplored. Modulation of miRNA levels in Mesenchymal Stem/Stromal Cells (MSC) is a novel approach to enhance their pro-regenerative properties and accelerate bone repair. We and others have shown that modulation of miRNAs can induce osteogenic differentiation and neovascularization, two crucial mechanisms for bone repair and regeneration. Also, we showed that cell construct priming in vitro promotes better in vivo outcomes.
miR4CMF aims to explore miRNA-engineered MSCs to modulate extracellular matrix (ECM) properties into a pro-regenerative, pro-osteogenic and pro-angiogenic profile, as an innovative solution to repair craniomaxillofacial fractures and defects. Specifically, it will: i) develop a 3D in vitro culture of miRNA-engineered MSC and ii) characterize the MSC newly synthetized pro-regenerative ECM, which in combination with the miRNA-induced MSC will work as seeding centers for new bone formation; iii) perform an in vivo proof-of-concept for the synergistic impact of miRNA-engineered MSC and their ECM in new bone formation, using the calvarial bone defect model.
miR4CMF will respond to the following scientific questions:
1. Are miRNAs useful tools to promote the synthesis and deposition of a pro-osteogenic and a pro-angiogenic bone ECM?
2. Can miRNA-engineered MSC and their synthetized ECM act synergistically to repair craniomaxillofacial fractures and defects?Using miRNA-based molecular engineering to promote an intracellular pro-osteogenic MSC profile, together with the synthesis of a pro-regenerative ECM, this project will offer a novel strategy to accelerate bone craniomaxillofacial regeneration. Importantly, this proposal opens a new line of investigation in the repair/regeneration of craniomaxillofacial fractures and defects.
-
AOCMFS-21-15L Project title: Releasing the "brakes" to drive tissue repair: targeting non-coding RNAs for bone regeneration
The treatment of large bone defects remains a major challenge for craniomaxillofacial (CMF) surgeons. Critical-sized bone defects are generally reconstructed with autologous bone grafts, but the procedure suffers from several disadvantages including limited tissue availability, donor-site morbidity and prolonged patient hospitalisation. This project aims to develop an effective strategy using mesenchymal stem cells (MSCs) enhanced by RNA therapy for the regeneration of large bone defects.
Cell therapy strategies using MSCs hold a great deal of promise for bone regeneration, but they have yet to become common clinical practice. A key factor contributing to this lack of progression is the inadequate vascularisation of MSC-based grafts, which can lead to necrosis of the constructs. There is increasing evidence that this challenge may be overcome by implanting chondrogenically-primed MSCs to induce endochondral ossification, the natural pathway of bone formation via a cartilage template. During this process, the MSC-derived cartilage induces the infiltration of blood vessels and remodelling into mature bone. Unfortunately, the chondrogenic priming of the cells requires extensive in vitro handling which severely hinders possible translation to the patient. In this project we will devise a new approach that will remove the need for prolonged in vitro pre-differentiation steps to induce MSC-mediated endochondral ossification in vivo. MSC commitment to the endochondral pathway will be achieved by blocking the activity of inhibitory non-coding RNAs (ncRNAs), regulatory molecules that can repress MSC differentiation. In the first phase of the project we will perform RNA-sequencing studies to unravel the changes in ncRNAs that occur in MSCs during the chondrogenic priming phase of endochondral ossification. This will lead us to identify key inhibitors of the process that can be targeted as molecular switches to trigger chondrogenesis and bone formation. Finally, we will establish a scaffold-based system to induce MSC-mediated endochondral ossification via delivery of anti-ncRNAs in situ, replacing the step of in vitro pre-differentiation. Ultimately, our study will provide proof-of-concept that blocking inhibitory ncRNAs in MSCs upon implantation removes the need for chondrogenic priming and leads to endochondral bone formation. This will bring tissue engineering strategies using MSCs and endochondral ossification closer to clinical application.
-
AOCMFS-20-21D Project Title: The Orbital Index: A Quantitative Tool for Prediction of Delayed Enophthalmos in Orbital Floor Fracture ManagementEarly identification of surgical indication is critical to optimizing outcomes in orbital floor fracture management. Delay of repair in this cohort risks persistent diplopia, persistent facial deformity, and infraorbital nerve injury. While muscle entrapment and acute globe malposition are widely accepted as absolute indications for repair, it has remained a challenge to identify those patients at risk for developing delayed enophthalmos and requiring subsequent surgery. The objective of this project is thus to validate a novel comprehensive quantitative clinically applicable prediction tool that guides orbital floor fracture management by stratifying risk for enophthalmos and establishing a threshold value for surgical intervention.
-
AOCMFS-20-16T Project title: Development of Virtual Reality based training in orthognathic surgery for CMF surgeonsThorough understanding of the spatial relationship of 3D anatomical structures is of paramount importance for physicians in general, and for surgeons operating on patients in particular. In craniomaxillofacial (CMF) surgery, the anatomy is complex and many important structures run in close proximity.
Nowadays, knowledge and understanding of these complex 3D shapes have to be learnt largely from images in books and screens, which lack the real 3D nature, and also do not permit interaction. It is well known that 3D interaction and immersion can contribute to a better understanding of objects around us. This accounts for all areas of the CMF surgery, but in particular for orthognathic surgery. The purpose of this project therefore, is to develop a Virtual Reality (VR)-based application that permits CMF residents to visualize and interact with facial skeleton in an immersive environment performing the LeFort I osteotomy and the bilateral sagittal split osteotomy (BSSO). More specifically, we will develop an application that allows them to: 1) provide insight in anatomical structures in the maxillary and mandibular region in a VR environment; 2) interact with these models by performing a leFort I osteotomy and a BSSO. While cutting through the bones the surgeons receive feedback from their handling via scoring. In this feedback, accuracy of the osteotomy and overall progress of handling can be reported.
There is a rapidly growing interest in the use of intelligent tools in virtual simulation of various applications for different training purposes. However, to the best of our knowledge there are none in the CMF-field. Aiming to simulate orthognathic surgery and effectively train clinicians to perform surgeries properly, we will develop in the VR-simulator for orthognathic surgery also a new tool to test the efficacy of the VR-simulator. The tool in the VR-simulator can provide the participants with real-time feedback to assist with training. The presented study aims to investigate evidence of validity of the tool while acquiring meaningful feedback regarding the potential use of the simulator and to get feedback from participants how they evaluate the use of VR in their training of orthognathic surgery. -
AOCMFS-20-05M Project title: MRRead – A Novel Approach to Training Residents in MRI TMJ InterpretationTemporomandibular disorders (TMD) are a common occurrence worldwide which can lead to a plethora of functional and emotional sequelae. Proper diagnosis of the direction, extent, and displacement of TMJ disorders by oral surgeons using the gold standard MRI TMJ Open and Closed views is imperative to improve patient outcomes and avoid unnecessary complications. Despite the importance of accurate diagnosis, oral surgery residents often receive a variable level of MRI interpretation training from a senior resident or staff in an unstructured manner. To address this inconsistency in training, this project aims to create a standardized, scalable training module called MRRead for all residents in oral and maxillofacial surgery so that they can become competent and confident when interpreting MRIs of the TMJ and presenting them to their fellow residents and staff. MRRead will be a web-based training module which will take a student, resident, or even staff member through the reading of a MRI scan one step at a time. Users will be shown normal MRI scans, taught how to interpret anatomy, and then taught how to identify common disorders in temporomandibular joints such as anterior disc displacement, joint effusion, osteoarthritis, etc. To test the effectiveness of MRRead, participants’ confidence level and actual interpretation skill will be assessed via a mandatory short but comprehensive survey and marked MRI interpretation quiz at the beginning and end of the training module. This will allow us to gather key information users’ confidence in interpreting MRI scans, and their ability to accurately diagnose traumatic fractures as they progress through the module. It is hoped that completion of MRRead will lead to a significant increase in both confidence level and actual interpretation skill level of oral surgery residents when it comes to reading MRI TMJs for patients.
-
AOCMFS-19-15K Project title: Osteoinductive potential of local administration of recombinant BMP9 in the bone defects in mice systemically treated with a monoclonal RANKL antibody drugThe monoclonal antibodies against Receptor Activator for Nuclear Factor Kappa-B Ligand (RANKL) such as denosumab, are used for the antibody mediated anti-resorptive therapies (AMARTs) in patients with metastatic cancer of the bone or osteoporosis. Bone augmentation procedures in the patients having AMARTs should be carefully considered due to the risk of antiresorptive agent-related osteonecrosis of the jaw (ARONJ). Therefore, the promising bone regeneration procedure is demanded for the patients with ARONJ risk.
In the past years, BMP9 has been characterized as one of most osteogenic bone-inducers among the BMP family. Our previous in vitro and in vivo reports revealed that recombinant human (rh)BMP9 demonstrated higher osteoinductive potential when compared to rhBMP2. Furthermore, our previous preliminary data interestingly showed positive effect of rhBMP2 on bone formation in mice after anti-murine monoclonal RANKL antibody (mAb) treatment. It is hypothesized that the local administration of rhBMP9 could further promote bone regeneration in animals having AMARTS. In this project, the recently commercially available mAb is used to create an AMART model in mice. Thereafter, rhBMP9 combined with collagen scaffold will be implanted in calvarial defects. After 4 weeks, the systemic effect of mAb and/or rhBMP9 treatment will be tested by measuring serum ALP level, TRAP-5b level and bone mineral density (BMD). The bone formation in the defects will be evaluated by microCT analysis, histomorphometry and immunohistochemical approach.
This project will show for the first time the effect of the rhBMP9 on bone regeneration potential in an AMART animal model. The results will contribute future bone regenerative therapy for the patients having AMARTS. -
AOCMFS-19-07N Project title: Trilineage differentiation potential of periosteal cells from endochondral and intramembranous originThe periosteum is a highly vascularized bilayer membrane covering the surfaces of bone. The outer “fibrous” layer consists of fibroblasts abundant amounts of extracellular components such as collagens and elastin giving stability and elasticity to the periosteum. In contrast, the inner “cambium” layer contains progenitor cells which are crucial for bone formation and repair. The periosteum exhibits osteogenic potential and has received considerable attention as a promising cell source for bone regeneration strategies. The high proliferation capacity and the simple availability compared to other sources of mesenchymal progenitor cells display big advantages as in vitro cell expansion is often a prerequisite for tissue engineered constructs. However, if and to which extent the harvest location affects the function of the periosteal derived cells is still unclear.
In the framework of this project we aim to compare the in vitro differentiation potential of human periosteum derived cells (hPDC) from endochondral and intramembranous origin. We hypothesize that differences in the developmental origin of the periosteum will influence the behavior and/or potency of hPDCs. We assume that due to the natural tendency for intramembranous ossification and the uniquely high bone remodeling occurring within the jaw, human JPDCs (jaw periosteal derived cells) represent a progenitor cell source with superior osteogenic properties. This would make hJPDCs more favorable for tissue engineering and regenerative medicine (TERM) of intramembranous bone.
This study is crucial for the characterization of hPDCs and can be used to guide clinical strategies that exploit periostea for tissue engineering and clinical applications. The present project may foster translational approaches for bone constructs which is of major interest for cranio-maxillofacial-, trauma- and orthopedic surgery. -
AO CMFS-18-25R Project title: Investigating effects of BMPER on osteogenic and chondrogenic differentiationBone morphogenetic proteins (BMPs) 2 and 7 have been approved for clinical use, yet complications limit their application. In addition, the supraphysiological doses applied would indicate that their use has not yet been optimized. BMP binding endothelial regulator (BMPER) also known as Crossveinless 2 was first identified as critical mediator in vein development (Conley, Silburn et al. 2000). Today it is known that BMPER is a BMP modulator, similar to Chordin, Noggin or Gremlin and interacts with BMP 2, 4, 6, 7, 9 and 10. In humans, the syndrome Diaphanospondylodysostosis (DSD) is caused by a lack / mutation of the BMPER protein. Characteristics are absent or severely delayed ossification of vertebral bodies and other bone defects, a short broad thorax, a short neck and respiratory insufficiency. The severe bone phenotype suggests that BMPER plays a major role in osteogenesis, yet it has been largely overlooked. No studies have been performed to assess whether BMPER as BMP modulator might be as osteoinductive as BMPs themselves nor whether BMPER is able to potentiate the osteoinductive effects of BMPs. The aim of our study is therefore to identify the osteogenic and chondogenic potential effects of BMPER and to establish whether BMPER is promising as a new factor for promoting bone and/ or cartilage regeneration. As a first step the effects of BMPER on MSCs (Mesenchymal stem cells) will be investigated in vitro.
-
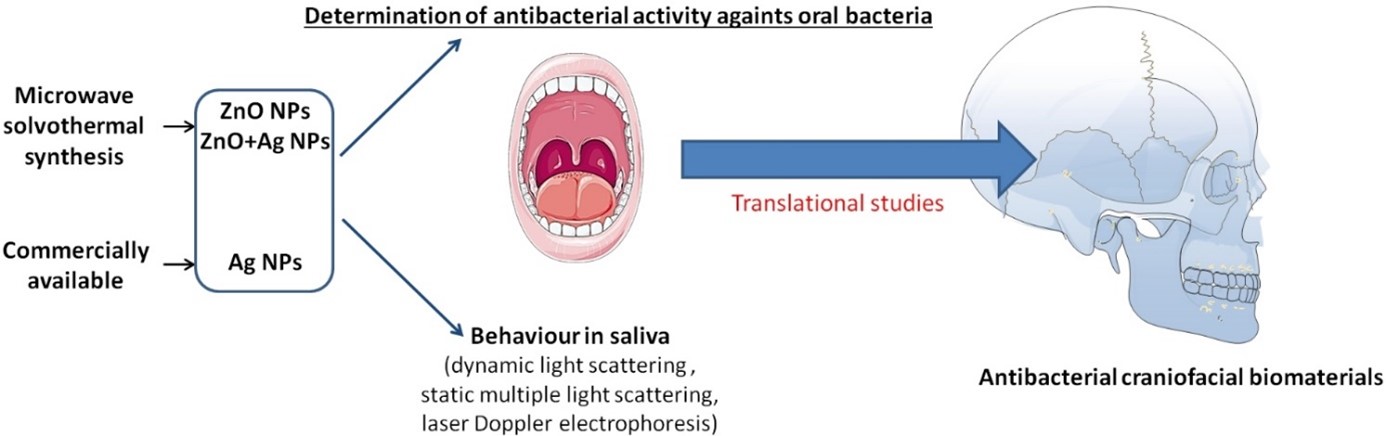
AO CMFS-18-14P Project title: Antibacterial nano-biomaterials for the purposes of cranio- maxillofacial surgeryInfections associated with implantable devices, also known as biomaterial associated infections (BAIs) pose a real problem in contemporary regenerative medicine and traumatology. In the head and neck area extraoral BAIs manifest as “pin sites infections” (PSI), while intraoral are known as peri-implant mucositis and/or peri-implantitis, which affects the underlying alveolar bone. Despite efforts in bioengineering to improve the biocompatibility of the metallic biomaterials, which constitute a major part of the reconstructive surgery, the problem of bacterial settlement and infection development still poses a serious threat for the treatment outcome. Along with nanotechnology evolution, antibacterial approaches with the use of different nanoparticles (NPs) were taken into concern. However, studies showed that such devices exhibited some limitations, mostly due to a restricted effective release rate, an initial burst release, cytotoxicity and unknown interactions of NPs with the host’s biomolecules. The aim of this study is twofold. First is to evaluate antibacterial activity of nano-sized zinc compounds against the bacteria responsible for infections around the biomaterials in the head and neck area. Second is to evaluate the stability of the nano-colloidal suspensions in the human and artificial saliva, and physico-chemical properties of the nano-particle-protein-sugar complexes, known as protein-coronas (PCs), in such environments which determine the activity of the NPs in the living organisms.
Published articles
2019 - Pokrowiecki R, Wojnarowicz J, Zareba T, Koltsov I, Lojkowski W, Tyski S, Mielczarek A, Zawadzki P. Nanoparticles and human saliva: a step towards drug delivery systems for dental and craniofacial biomaterials. International Journal of Nanomedicine (Future Medicine). 2019:14, 9235-9257 Manuscript number NNM-2019-0189 (original article). doi: 10.2147/IJN.S221608
Pokrowiecki R. The paradigm shift for drug delivery systems for oral and maxillofacial implants. Drug Deliv. 2018 Nov;25(1):1504-1515. doi: 10.1080/10717544.2018.1477855.
Book chapter
Palka K, Pokrowiecki R, Krzywicka M. Chapter 13: Porous titanium materials and applications in “Titanium for Consumer Applications” Book. 1st Ed. ISBN: 9780128158203. Elsevier, 2019.
-
AO CMFS-18-02M Project title: CTRead - A revolutionary approach to training residents in CT Facial Bones interpretation
Facial trauma is a common occurrence worldwide which can lead to a plethora of functional, cosmetic, and emotional sequelae. Proper diagnosis of the direction, extent, and displacement of facial fractures by oral surgeons using the gold standard high-resolution CT scanning is imperative to improve patient outcomes and avoid unnecessary complications. Despite the importance of accurate diagnosis, oral surgery residents often receive a variable level of CT interpretation training from a senior resident or staff in an unstructured manner. To address this inconsistency in training, this project aims to create a standardized, scalable training module called CTRead for new residents in oral and maxillofacial surgery everywhere so that they can become competent and confident when interpreting scans and presenting them to their senior residents and staff. CTRead will be a web based training module which will take a student, resident, or even staff member through the reading of a CT Facial Bones scan one step at a time. Users will be shown normal CT scans, taught how to interpret anatomy, and then taught how to identify common fractures in facial trauma patients. To test the effectiveness of CTRead, participants’ confidence level and actual interpretation skill will be assessed via a mandatory short but comprehensive survey and marked CT interpretation quiz at the beginning and end of the training module. This will allow us to gather key information users’ confidence in interpreting CT scans, and their ability to accurately diagnose traumatic fractures as they progress through the module. It is hoped that completion of CTRead will lead to a significant increase in both confidence level and actual interpretation skill level of oral surgery residents when it comes to reading CT Facial Bones scans for trauma patients.
Published articles
Mascarenhas W, Richmond D, Chiasson G. CTRead-A Revolutionary Approach to Training Residents in Computed Tomography Facial Bone Interpretation.
J Oral Maxillofac Surg. 2019 Apr 23. pii: S0278-2391(19)30447-1. doi: 10.1016/j.joms.2019.04.016. [Epub ahead of print] -
AO CMFS-17-20K Project title: Fully MRI based 3D virtual planning of CMF tumour resection and free flap reconstruction
The aim of the proposed clinical study is to change the conventional CT-based 3D virtual planning workflow for CMF tumors by developing a method for MRI-based tumor resection as well as free flap reconstructive planning. In addition the MRI-based workflow is complemented with the quantification of vascular flow in both donor and recipient site, as well as the 3D visualization of perforators. Project description: Multiple types of MRI sequences will be studied for visualizing bone and vascularization. Several MRI- sequences are known to be suitable for segmenting bone for 3D planning, these will be explored within this project. Also MR-Angiogram with flow measurements will be selected for visualizing arteries and to quantify the arterial flow, thereby determining suitability for transplantation. A workflow for MRI-based 3D surgical planning with bone cutting guides will be developed using a four-step approach. Key MRI parameters are defined (phase 1), followed by an application of selected bone and Phase Contrast-MRI sequences on healthy volunteers (phase 2).The most suitable (bone and Phase Contrast) MRI sequences will be chosen for phase 3. These protocols are validated by applying them on patients (n=10) and comparison to corresponding patients CT data, which is the gold standard. The mean deviation values between the MRI- and the CT-based models are determined by 3D comparison analysis as a primary outcome measure.Already for validation of Black Bone sequences approval has been obtained from the local medicalethical board (file number M16.198347).
Phase 4 entails examination of the clinical value during surgery and in pre-op clinical decision making,using bone cutting guides (for mandible/maxilla and fibula) designed from MRI-based models, inpatients with oral cancer who will undergo surgical treatment with free flap reconstruction. Secondary outcome measures are 1) the deviation of the actual bone cutting planes in the CMF region as well asat the bone-free flap, compared to the 3D planning and 2) the fit of the guides to the bone surface. The final result of this project is aimed to be a complete MRI based 3D virtual planning workflow including resection of the tumor and free flap reconstruction planning.Published articles
Optimisation of three-dimensional lower jaw resection margin planning using a novel Black Bone magnetic resonance imaging protocol.
PLoS One. 2018 Apr 20;13(4):e0196059. doi: 10.1371/journal.pone.0196059. eCollection 2018. -
AO CMFS-17-15D Project title: Creation and validation of a 3D cephalometric software based on Delaire’s analysis
There is a large number of cephalometric analyzes, based on the analysis of facial points or based on radiology analyzes. Nowadays new technologies allow us to catch and analyze patients’ faces (thanks to 3D photography) and to get simple access to head CT-scans. Under those circumstances, our cephalometric analyzes should evolve in order to adapt to these features.
There has already been attempts to create 3D cephalometric analyzes based on CT-scans, but none of them were based on a dedicated software. Therefore results were impossible to reproduce. We propose to create a 3D cephalometric analysis free software for research based on CT-scans DICOM files, easy to use and available for everyone.
Our first step after the creation of the software is to plan a comparative study between “classic” radiography analysis and 3D analysis, on a cohort of 100 to 150 patients with a point to point comparison, to show that the 3D analysis is theoriticaly at least as good as the classical radiographic one.
The advantages of this new technology would be a greater precision in cephalometric outline, and so in the analysis of the patient facial lines. By analyzing a 3D volume we also intend to answer some multi-dimensional questions about the chin position, mandibular asymmetry and other complex facial skull movements.