New Templates for Universal Small Fragment System
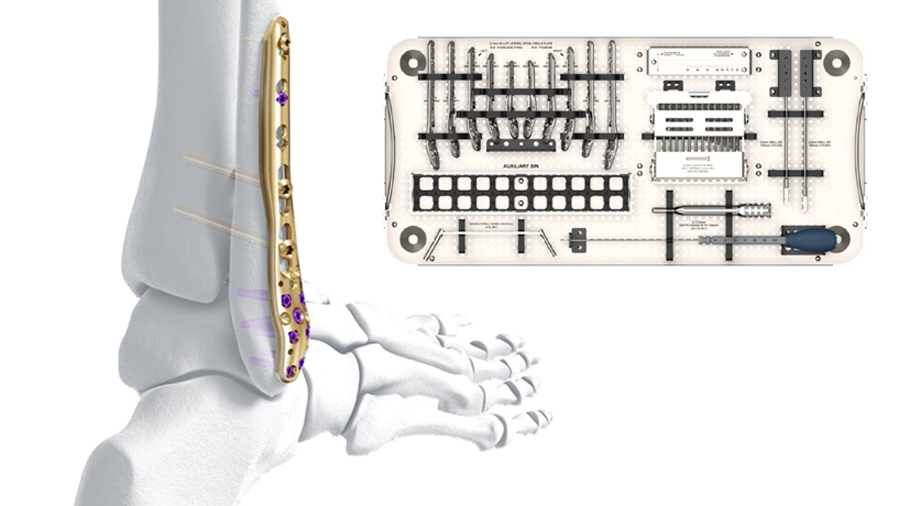
In 2019, the Universal Small Fragment (USF) System was launched and consists of a core set and eight streamlined modular anatomic implant trays to support small fragment procedures with non-locking, locking, and variable angle locking plate technologies. To enable proper implant selection, we are pleased to report that new templates complement all anatomic plate types of the USF System.
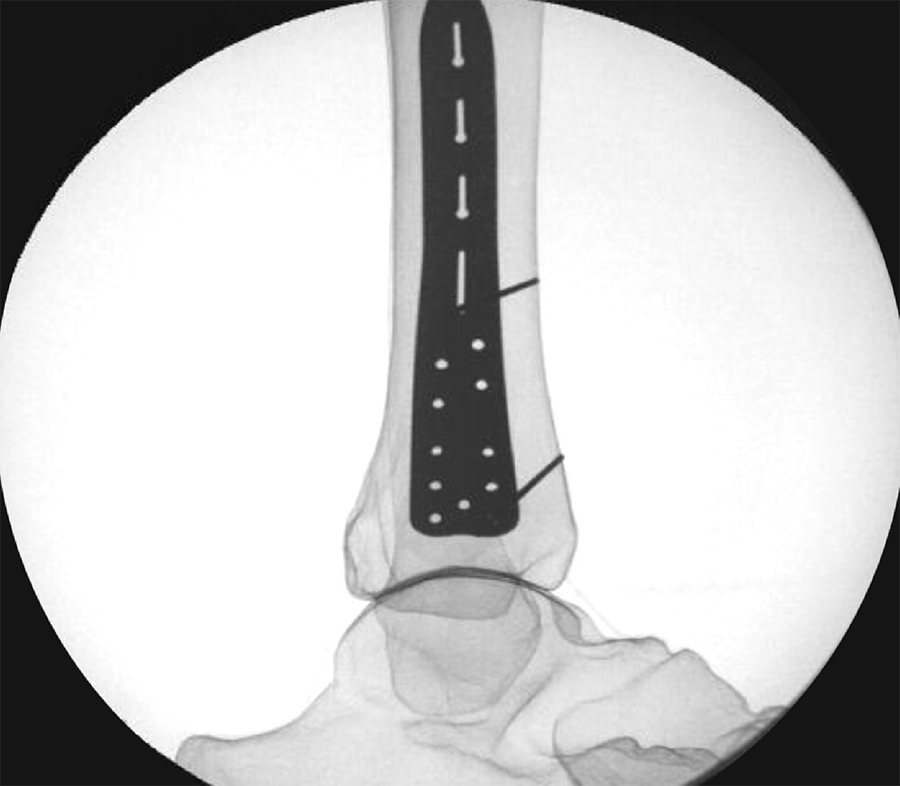
To provide an economical and effective solution, the new templates are manufactured using 3D printing technology and utilizing non-implantable stainless steel. The templates mimic the shape of the respective anatomic plates (Fig 1) and allow reprocessing for repeated use. They are radiopaque, and K-wire holes indicate the location of plate holes (Fig 2). To minimize system complexity, templates are provided for the shortest and most common length of an implant. Determination of other plate sizes can be deduced from straight measurements. The templates are labeled “DO NOT BEND” as they will no longer mimic the partnered implant, and repeated bending can result in template breakage.
The following plate families are supported by the templates:
Upper extremity:
- Proximal humerus: LCP® Proximal Humerus (PHILOS) and LCP® Periarticular Proximal Humerus
- Clavicle: LCP® Superior Anterior and Superior Clavicle (with extension and standard), VA LCP® Anterior Clavicle
- Elbow: VA LCP® Medial Distal Humerus, VA LCP® Lateral Distal Humerus, VA LCP® Posterolateral Distal Humerus, VA LCP® Proximal Olecranon, VA LCP® Olecranon, LCP® Extra-articular Distal Humerus, LCP® Hook
Lower extremity:
- Proximal Tibia: VA LCP® Proximal Tibia (small and large bend), LCP® Proximal Tibia (standard and low bend), LCP® Posteromedial Proximal Tibia, LCP® Medial Proximal Tibia
- Distal Tibia: VA LCP® Medial Distal Tibia, VA LCP® Anterolateral Distal Tibia, LCP® Anterolateral Distal Tibia, VA LCP® Posterolateral Distal Tibia, VA LCP® Posterolateral Distal Tibia T-Plate, LCP® Low Bend Medial Distal Tibia
- Distal Fibula: VA LCP® Lateral Distal Fibula, LCP® Lateral Distal Fibula, LCP® Hook
The new templates will be made available worldwide in a sequential manner beginning early 2021. Japan, Germany, the United Kingdom, and the Nordic countries are among those markets where the templates will be released first.
Disclaimer:
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
The brands and labels "approved by AO Technical Commission" and "approved by AO Foundation", particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.