DePuy Synthes Hammertoe Continuous Compression Implant
Hammertoe deformities are one of the most frequent deformities treated by foot and ankle surgeons. In rigid and structured hammertoe deformities not suited for nonoperative management, arthrodesis of the proximal interphalangeal (PIP) joint represents the standard treatment. Temporary K-wire fixation is the most frequently used, low-cost fixation method for PIP joint fusion. However, reported complications associated with K-wires have prompted the development of new implants over the past decade. To address weaknesses of intramedullary fixation concepts (eg, limited rotational stability), the Foot and Ankle Expert Group approved the first extramedullary continuous compression implant (CCI) to treat hammertoe deformities – the DePuy Synthes Hammertoe Continuous Compression Implant (DPS Hammertoe CCI).
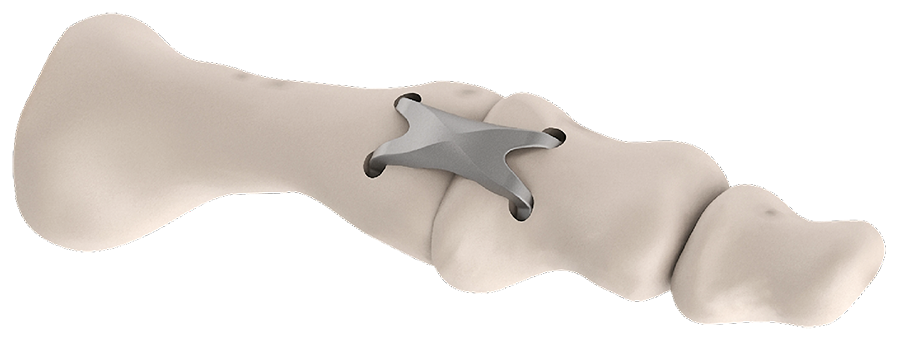
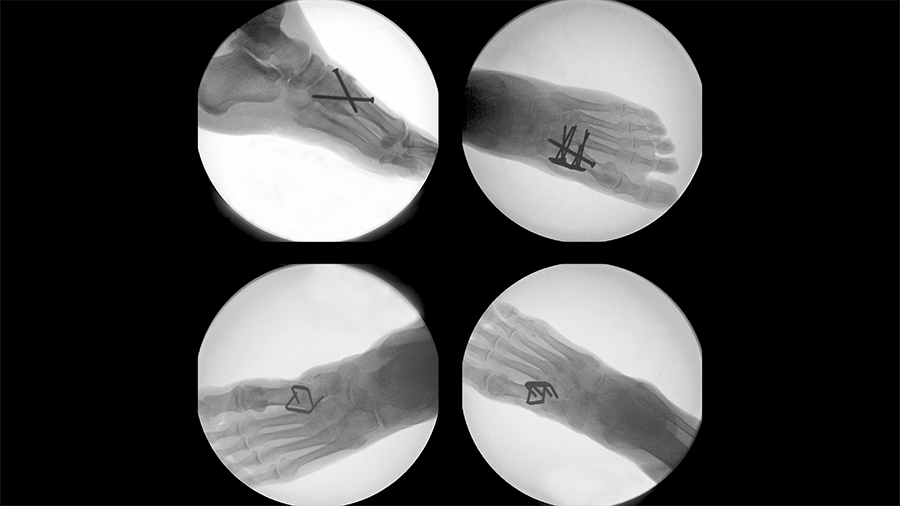
The DPS Hammertoe CCI has a staple-like design with four legs and a low profile connecting bridge (Fig 1). It is made of biocompatible Nitinol, a metal alloy of nickel and titanium, which is known for its superelastic properties and shape memory behavior.
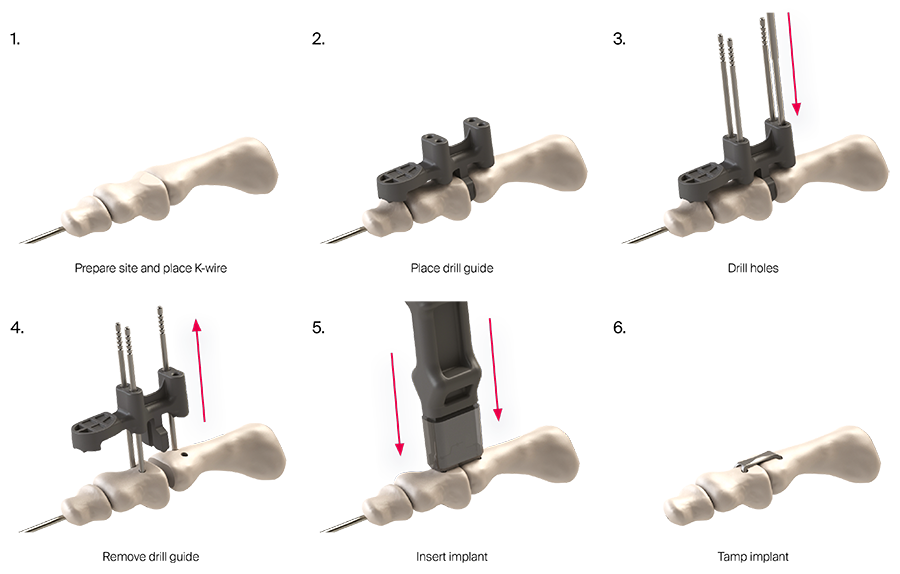
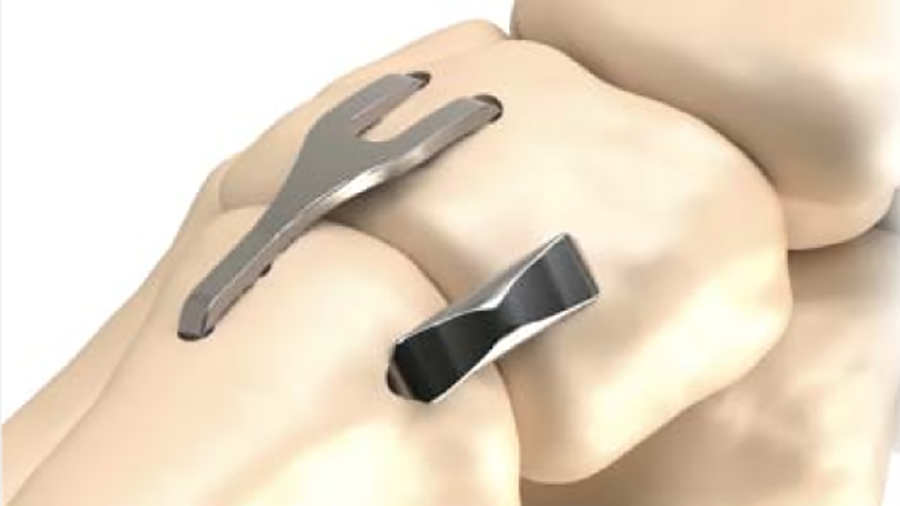
The implant system is delivered to the operating room in a disposable, sterile kit containing the Insertion Stick with the pre-loaded implant (Fig 2) and other instruments needed for the implantation procedure (Fig 3).
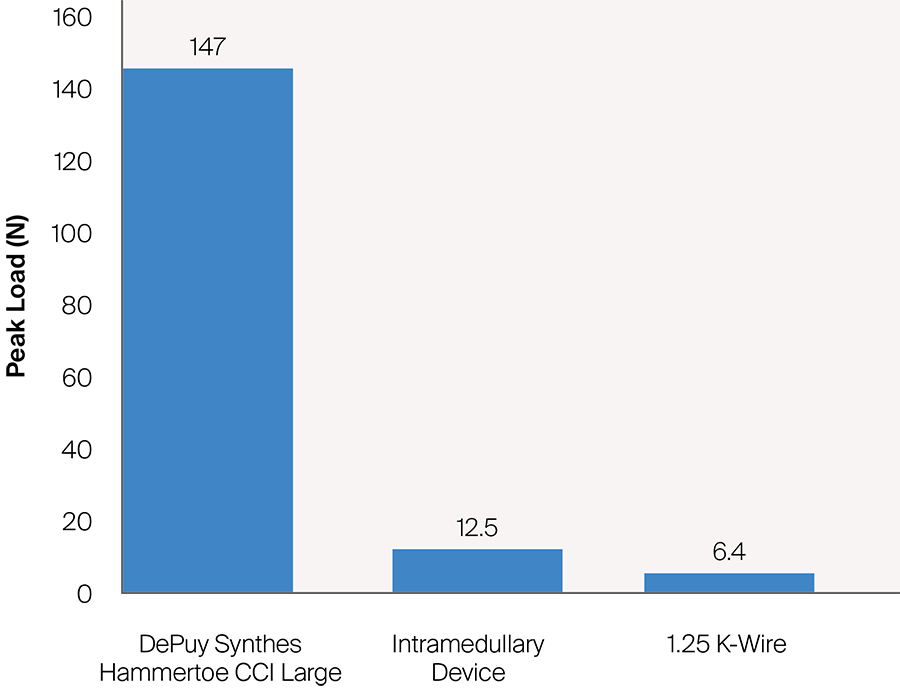
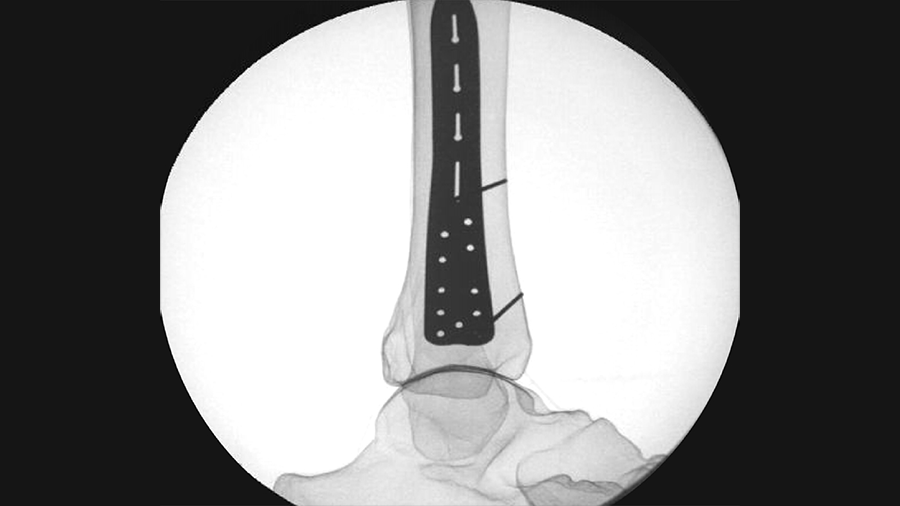
Once the DPS Hammertoe CCI is inserted into the bone and released from the Insertion Stick, the implant attempts to regain its original shape with converging legs thereby providing active continuous compression at the fusion site (Fig 4). The design with the four legs orthogonal to the bone axis and the active compression feature are beneficial for the distraction resistance (Fig 5).
The implant is not supposed to be reused after it has been discharged from the Insertion Stick because any processing, reprocessing, or mechanical manipulation may reduce the effectiveness of the implant.
So far, the DePuy Synthes Hammertoe CCI System is only available in the United States. However, it is under consideration for introduction to other regions of the world.
Disclaimer:
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
The brands and labels "approved by AO Technical Commission" and "approved by AO Foundation", particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.