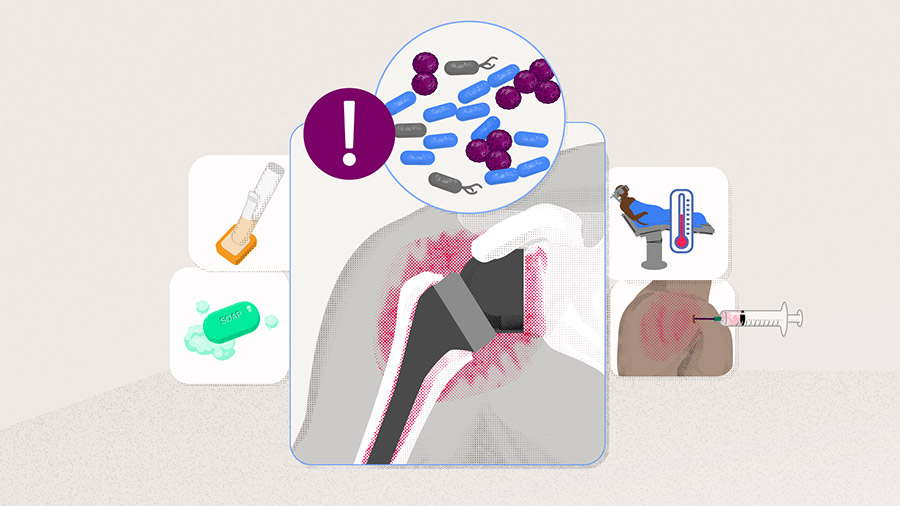
Preventing infection in reverse shoulder arthroplasty
Preview
Reverse shoulder arthroplasty (RSA) is a highly effective and generally safe procedure. However, periprosthetic joint infections (PJI) occur more frequently than in anatomical shoulder arthroplasty [1]. Although rare, they have the potential to significantly increase the short- and long-term health risks. Revision surgery is often required and commonly has a substantially poorer outcome than the primary procedure [2]. It is imperative that a surgical team works to prevent, recognize, treat, and monitor infection if it does arise—prevention is key. In this article Simon Lambert, an experienced consultant orthopedic and trauma surgeon at the University College London Hospital, UK, gives an overview of steps to follow to prevent infection before, during, and after RSA surgery.

Simon Lambert
AO Recon Shoulder Curriculum Taskforce
University College London Hospital
London, United Kingdom
Periprosthetic joint infection: What should we be prepared for?
The most common pathogens causing periprosthetic joint infection (PJI) are the gram-positive coccus Staphylococcus (S ) aureus, the family of coagulase-negative staphylococci, specifically S epidermidis, and Cutibacterium acnes (C acnes) [3–5], which has the ability to evade rigorous skin preparation and administered prophylactic antibiotics [6, 7]. Cutibacterium acnes is becoming more problematic in postoperative shoulder infections and is elusive, requiring particular laboratory culture strategies to diagnose its presence [8]. Understanding the types of bacteria that are most frequent in this type of surgery can help the surgical team to prepare for and eliminate them in the operating room setting.
Being vigilant throughout the whole procedure: pre-, peri- and postoperative steps to follow
The incidence of infection with RSA varies between 0.3% and 6% [4, 9]. This increases further after revision RSA [7, 10] and also following RSA performed after previous surgical interventions. As the aging population increases in number, so will the number of RSAs. Reducing the risk of infection associated with RSA relies on detailed preoperative preparation, refining surgical exposure techniques to reduce surgical operating time and intraoperative bleeding, and limiting the incidence of postoperative adverse events (AEs), particularly bleeding and primary wound healing problems. The medical team can take several measures to reduce the risk of opportunistic bacteria causing nosocomial infection.
Preparation of the patient
The treating surgeon should fully understand the general health of their patient and, if needed, optimize their health before the procedure so as to lower the risk of infections and complications. Comorbidities including the related conditions of bleeding disorders (eg, intrinsic coagulopathies, liver disease, anemia of all causes), microvascular diseases (diabetes, lupus erythematosus), immunosuppressive conditions and treatments (eg, the inflammatory arthritides, ongoing corticosteroid therapy, immunosuppressive therapies), and disorders characterized by poor metabolite handling (eg, renal failure, malnutrition) have been reported as risk factors for infection [3, 7]. Other modifiable factors, such as high body mass index, smoking, and excessive alcohol use, increase the risk of PJI [11]. Surgeons should evaluate these comorbidities preoperatively, obtain optimal advice from colleagues regarding the cessation of medications, and/or bridging of disease treatments during the perioperative period, and ensure that the patient is as fit as reasonably possible before undergoing surgery.
To give an example of the available literature, Cancienne et al [9], for instance, analyzed infection and risk factors in a large study Medicare population in the US and reported a rate of 0.26% for infections within 90 days after RSA [9]. Revision surgery (after any other intervention) was the most significant risk factor for infection, while intraoperative steroid injection was an independent risk factor for postoperative infection. The study highlighted that obesity, male gender, chronic anemia, malnutrition, depression, and alcohol use were all patient-related risk factors for postoperative infection [9]. Previous intraarticular corticosteroid injections administered for the management of rotator cuff disease, previous surgical interventions (whether arthroscopic or open) including prior rotator cuff surgery and arthroplasty are all associated with an increased risk of PJI, particularly in RSA [11].
Read the full article with your AO login
- Specific issues and diagnostic criteria
- Preparation of the skin and anesthesia
- Intraoperative strategies
- The role of antibiotics
- The role of bleeding and hermatoma
- Antibiotic-loaded cement
- Topical antiseptics
- Wound closure
- Postoperative strategies
- Treatment algorithm
- Conclusion
Additional AO resources
Access videos, tools, and other assets.
- Videos
- Upcoming events: AO Recon Course finder
Contributing experts

Ashish Gupta
Queensland Orthopedic Clinic
Brisbane, Australia

Simon Lambert
AO Recon Shoulder Curriculum Taskforce
University College London Hospital
London, United Kingdom

Markus Scheibel
AO Recon Shoulder Curriculum Taskforce
Schulthess Clinic
Zurich, Switzerland

Jan-Philipp Imiolczyk
Charité—University Medicine
Berlin, Germany
The authors thank Antia Rodriguez-Villalon and Laura Kehoe, medical writers at AO Innovation Translation Center, Switzerland, for contributing to the writing and editing of the articles.
References
- Contreras ES, Frantz TL, Bishop JY, et al. Periprosthetic Infection After Reverse Shoulder Arthroplasty: a Review. Curr Rev Musculoskelet Med. 2020 Dec;13(6):757–768.
- Boileau P. Complications and revision of reverse total shoulder arthroplasty. Orthop Traumatol Surg Res. 2016 Feb;102(1 Suppl):S33–43.
- Nelson GN, Davis DE, Namdari S. Outcomes in the treatment of periprosthetic joint infection after shoulder arthroplasty: a systematic review. J Shoulder Elbow Surg. 2016 Aug;25(8):1337–1345.
- Pulido L, Ghanem E, Joshi A, et al. Periprosthetic joint infection: the incidence, timing, and predisposing factors. Clin Orthop Relat Res. 2008 Jul;466(7):1710–1715.
- Topolski MS, Chin PY, Sperling JW, et al. Revision shoulder arthroplasty with positive intraoperative cultures: the value of preoperative studies and intraoperative histology. J Shoulder Elbow Surg. 2006 Jul-Aug;15(4):402–406.
- Saltzman MD, Marecek GS, Edwards SL, et al. Infection after shoulder surgery. J Am Acad Orthop Surg. 2011 Apr;19(4):208–218.
- Bonnevialle N, Dauzères F, Toulemonde J, et al. Periprosthetic shoulder infection: an overview. EFORT Open Rev. 2017 Apr;2(4):104–109.
- Clark JJC, Abildgaard JT, Backes J, et al. Preventing infection in shoulder surgery. J Shoulder Elbow Surg. 2018 Jul;27(7):1333–1341.
- Cancienne JM, Brockmeier SF, Carson EW, et al. Risk Factors for Infection After Shoulder Arthroscopy in a Large Medicare Population. Am J Sports Med. 2018 Mar;46(4):809–814.
- Foruria AM, Fox TJ, Sperling JW, et al. Clinical meaning of unexpected positive cultures (UPC) in revision shoulder arthroplasty. J Shoulder Elbow Surg. 2013 May;22(5):620–627.
- Garrigues GE, Zmistowski B, Cooper AM, et al. Proceedings from the 2018 International Consensus Meeting on Orthopedic Infections: management of periprosthetic shoulder infection. J Shoulder Elbow Surg. 2019 Jun;28(6s):S67–s99.
- Friedman DJ, Parnes NZ, Zimmer Z, et al. Prevalence of cerebrovascular events during shoulder surgery and association with patient position. Orthopedics. 2009 Apr;32(4).
- Van Tongel A, Hardeman F, Karelse A, et al. Positioning of the patient during shoulder surgery: an inexpensive, safe and easy technique. Eur J Orthop Surg Traumatol. 2013 Jan;23(1):115–117.
- Saltzman MD, Nuber GW, Gryzlo SM, et al. Efficacy of surgical preparation solutions in shoulder surgery. J Bone Joint Surg Am. 2009 Aug;91(8):1949–1953.
- Ackermann W, Fan Q, Parekh AJ, et al. Forced-Air Warming and Resistive Heating Devices. Updated Perspectives on Safety and Surgical Site Infections. Front Surg. 2018;5:64.
- Kümin M, Deery J, Turney S, et al. Reducing Implant Infection in Orthopaedics (RIIiO): Results of a pilot study comparing the influence of forced air and resistive fabric warming technologies on postoperative infections following orthopaedic implant surgery. J Hosp Infect. 2019 Dec;103(4):412–419.
- Verra WC, Beekhuizen SR, van Kampen PM, et al. Self-Warming Blanket Versus Forced-Air Warming in Primary Knee or Hip Replacement: A Randomized Controlled Non-Inferiority Study. Asian J Anesthesiol. 2018 Dec;56(4):128–135.
- Chataule SM, Hazarika A, Jain K, et al. Preoperative Forced-Air Warming Strategy: Is It Effective in Averting Intraoperative Hypothermia in Elderly Trauma Surgical Patients? Cureus. 2022 Sep;14(9):e29305.
- Tjoakarfa C, David V, Ko A, et al. Reflective Blankets Are as Effective as Forced Air Warmers in Maintaining Patient Normothermia During Hip and Knee Arthroplasty Surgery. J Arthroplasty. 2017 Feb;32(2):624–627.
- Scholten R, Leijtens B, Hannink G, et al. General anesthesia might be associated with early periprosthetic joint infection: an observational study of 3,909 arthroplasties. Acta Orthop. 2019 Dec;90(6):554–558.
- Scholten R, Leijtens B, Kremers K, et al. The incidence of mild hypothermia after total knee or hip arthroplasty: A study of 2600 patients. J Orthop. 2018 Jun;15(2):408–411.
- Marigi EM, Bartels DW, Yoon JH, et al. Antibiotic Prophylaxis with Cefazolin Is Associated with Lower Shoulder Periprosthetic Joint Infection Rates Than Non-Cefazolin Alternatives. J Bone Joint Surg Am. 2022 May 18;104(10):872–880.
- Oprica C, Nord CE. European surveillance study on the antibiotic susceptibility of Propionibacterium acnes. Clin Microbiol Infect. 2005 Mar;11(3):204–213.
- Lovallo J, Helming J, Jafari SM, et al. Intraoperative intra-articular injection of gentamicin: will it decrease the risk of infection in total shoulder arthroplasty? J Shoulder Elbow Surg. 2014 Sep;23(9):1272–1276.
- Nowinski RJ, Gillespie RJ, Shishani Y, et al. Antibiotic-loaded bone cement reduces deep infection rates for primary reverse total shoulder arthroplasty: a retrospective, cohort study of 501 shoulders. J Shoulder Elbow Surg. 2012 Mar;21(3):324–328.
- Cheung EV, Sperling JW, Cofield RH. Infection associated with hematoma formation after shoulder arthroplasty. Clin Orthop Relat Res. 2008 Jun;466(6):1363–1367.