TriALTIS™ Spine System
Christian Mazel, Naresh Kumar, Firoz Miyanji, Yu-Mi Ryang, Kevin Seex
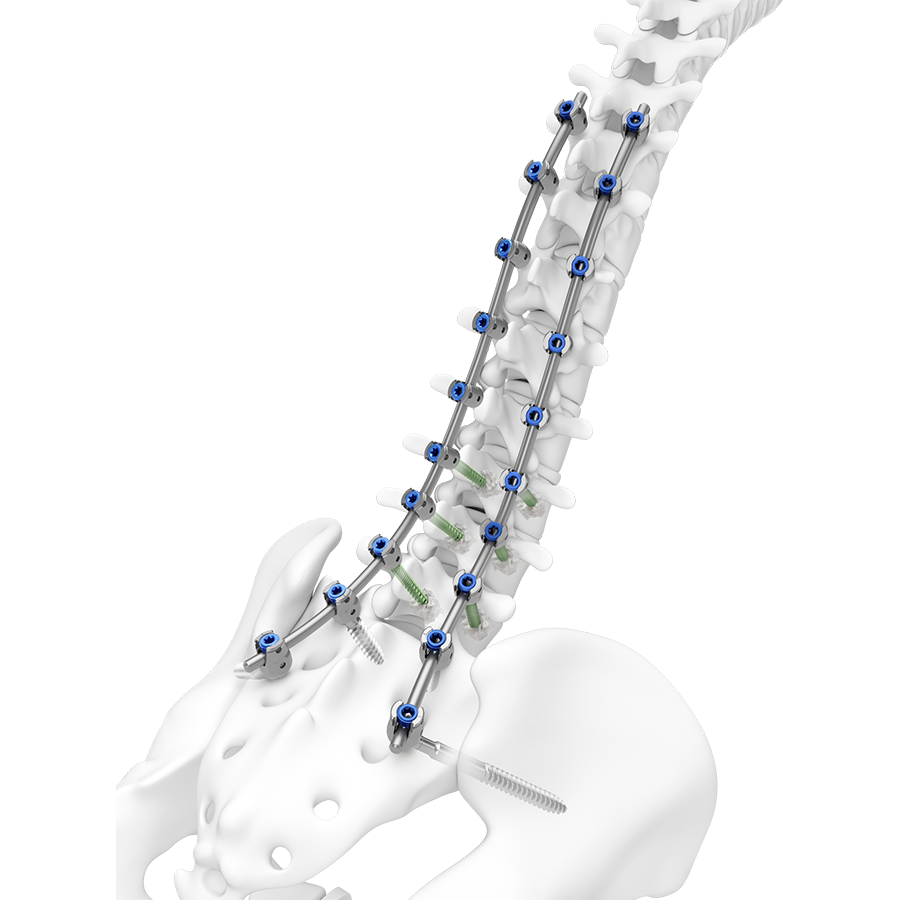
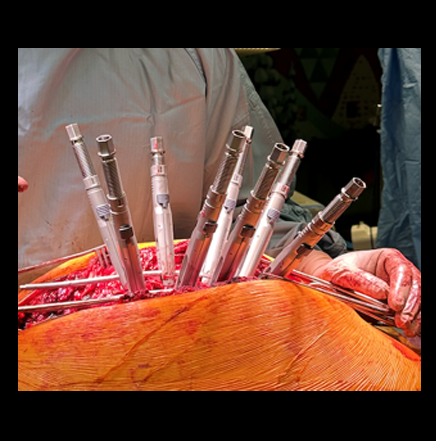
The TriALTIS™ Spine System (Fig 1) is a next generation posterior thoracolumbar spine implant system with a clinical focus on open surgery for degenerative, tumor, trauma and deformity pathologies in adults. Combining a new portfolio of implants with digital enabling technology, the TriALTIS™ Spine System aims to address unmet clinical needs and help surgeons achieve more consistent outcomes in treating complex spine conditions. Novel implants include polyaxial screws, favored angle screws, set screws, rods, and instruments.
Leveraging design features of the EXPEDIUM™ 5.5, VIPER™ Spine, and MATRIX™ Spine Systems, the TriALTIS™ Spine System delivers a consistent user experience and improved implant performance in terms of set screw clamping force, set screw loosening torque, resistance to pullout, and gripping capacity. Other innovations include improved instrument design to indicate actuation points, consolidated instrument sets, and a modular case and tray design.
The TriALTIS™ system affords efficient integration with cement augmentation, power, navigation and robotic-assisted solutions, while decreasing costs to healthcare systems and addressing the needs of more patients. The TriALTIS™ Spine System is the foundational element of a wider comprehensive system (coming soon) that will allow for a consistent surgeon user experience across all indicated pathologies.
Approved in 2025, the TriALTIS™ Revision System is a complementary extension to the original TriALTIS™ Spine System and comprises a new module of rod connectors and surgical instruments.
TriALTIS™ Implant Portfolio
Poly screws
Poly screws are available in diameters ranging from 4.5–7.0 mm (Fig 2). Solid cortical fix screws are available in lengths from 20–60 mm, and cannulated fenestrated cortical fix screws are available in lengths from 30–60 mm.
Rods
The TriALTIS™ Spine System offers an increased availability of rod options and materials for various clinical applications and anatomies. Rods are available in both Ti and CoCr, in diameters of 5.5 mm and 6.0 mm. The 6.0 mm rod option has improved fatigue performance compared to 5.5 mm rods of the same material and may be advantageous in deformity correction. The TriALTIS™ system is compatible with Symphony™ OCT and Altalyne™ rods.
TriALTIS™ pedicle screws
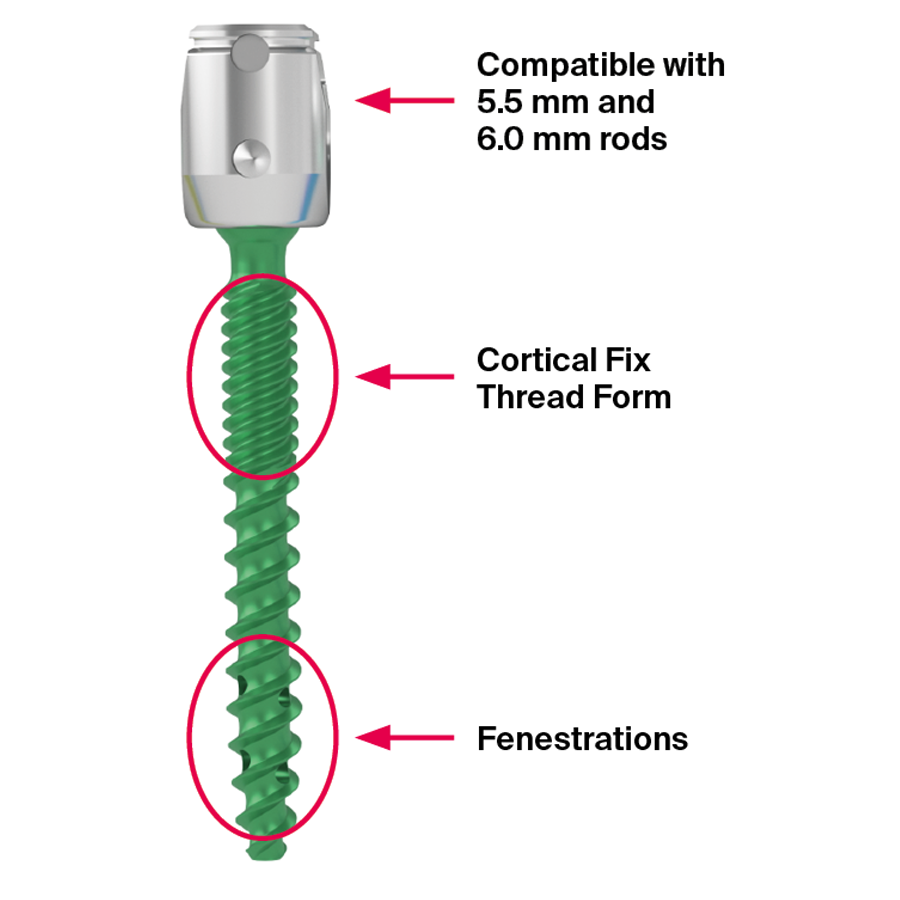
Design features (Fig 3) include:
- 5.5/6.0 mm diameter rod compatibility.
- Cortical fix thread form: this feature is standard on all TriALTIS polyaxial screws and available in solid and fenestrated screws.
- Fenestrations for cement augmentation: the injection of bone cement through the screw allows additional fixation in patients with advanced stage tumors involving the thoracic and lumbar spine.
- Polyaxial drag: the screw head features an interference fit between the shank sphere/flex ball and the drag ring. This friction fit allows the polyaxial heads to maintain their user designated position in-situ [1].
- Quick catch tip: allows for immediate bone engagement. This design feature consists of a bullet-shaped screw tip that allows the screw threads to extend all the way to the distal end, meaning that the effective diameter of the start of the threads is larger. TriALTIS™ screws offer improved starting efficiency when compared to the EXPEDIUM™ 5.5 Spine System [2].
The unique design features of the TriALTIS™ cortical fix screw (Figs 3 and 4) allow the most challenging clinical cases to be addressed:
- The cortical fix thread form doubles the number of contact points within the pedicle and increases the resistance to axial pull out forces, ultimately enhancing fixation within the pedicle.
- The dual lead thread integrates with the cancellous bone in the vertebral body and advances twice as far with each rotation (as compared to single lead screws).
- Additional proximal threads engage more of the posterior wall of the pedicle.
- The constant dual thread lead gives a smooth transition between the dual lead and quad lead thread forms, allowing for ease of insertion and compatibility with TriALTIS™ system taps.
- The optimized self-tapping and self-centering tip allows for insertion with or without tapping.
Mechanical testing shows that TriALTIS™ cortical fix thread form screws have 24% increased pull out strength compared to EXPEDIUM dual lead thread form screws [3].
In terms of overall construct stability, TriALTIS™ Screws demonstrated statistically higher gripping capacity [4,5] and statistically higher performance in cantilever beam testing [6,7] when compared to EXPEDIUM VERSE Screws.
TriALTIS™ set screws
Design features include:
- Star drive recess offering improved driver strength with the TriALTIS™ Spine System’s T27 driver.
- The set screw thread features a square cross-sectional geometry and a coarse thread pitch, offering a significant improvement over standard threads and reducing the propensity for cross-threading of the implants during insertion. The distance between threads is 1.4 mm for TriALTIS set screws compared to 1.0 mm for Expedium 5.5 screws [8].
Mechanical testing shows that compared to the EXPEDIUM 5.5 Spine System, the TriALTIS™ set screw offers a 20% improvement in set screw loosening torque and a 16% increase in set screw clamping force [9].
Clinical needs addressed
- The TriALTIS™ Spine System is designed to meet the demands of varying patient populations
- The streamlined system offers improved procedural efficiency with 5.5/6.0 mm diameter rod compatibility
- The system offers improved mechanical performance which may lead to reduced postoperative mechanical failure
- The system offers efficient integration with power and navigation systems
References
1. DePuy Synthes. Screw Polyaxial Drag. September 25, 2023. Windchill Document No. 501360165 Rev A.
2. DePuy Synthes. TriALTIS Quick Catch and Self-Tapping Analysis. September 05, 2023. Windchill Document No. 501380612, Rev A.
3. DePuy Synthes. TriALTIS Screw Pull-Out Comparison. September 05, 2023. Windchill Document No. 501365371, Rev A.
4. DePuy Synthes. TriALTIS System Poly Axial Slip Testing. December 12, 2023. Windchill Document No. 501452611 Rev A.
5. DePuy Synthes. TriALTIS System LDFA Axial Slip Testing. December 12, 2023. Windchill Document No. 501452681 Rev A.
6. DePuy Synthes. TriALTIS System Poly Static Cantilever Beam Testing. December 12, 2023. Windchill Document No. 501452609 Rev A.
7. DePuy Synthes. TriALTIS System LDFA Static Cantilever Beam Testing. December 12, 2023. Windchill Document No. 501452678 Rev A.
8. DePuy Synthes. Implant Cross Threading Memo. September 05, 2023. Windchill Document No. 501337048 Rev A.
9. DePuy Synthes. TriALTIS System Torque and Clamping Comparison to Predicates. September 05, 2023. Windchill Document No. 501335337, Rev A.
Instruments
The consolidated instrument sets within the TriALTIS™ Spine System include different types of reducers, including the sequential reducer, rocker reducer and acute reducer. The sequential reducer is shown in Fig 5.
Integration with enabling technology
Throughout the TriALTIS™ system, instruments are designed with standardized features to allow for consistent interaction with enabling technology. Instrumentation for navigation integration includes drill bits, taps, and screwdrivers:
- Navigated taps range from 3.5 mm–12.0 mm diameter
- Navigated drill bits come in 2.4 mm, 3.2 mm, and 4.0 mm diameter
Instrumentation also includes drill sleeves that enable one-handed adjustment of the navigation array.
Clinical cases kindly provided by Firoz Miyanji, MD, Department of Orthopedics, University of British Columbia, Canada
Case 1
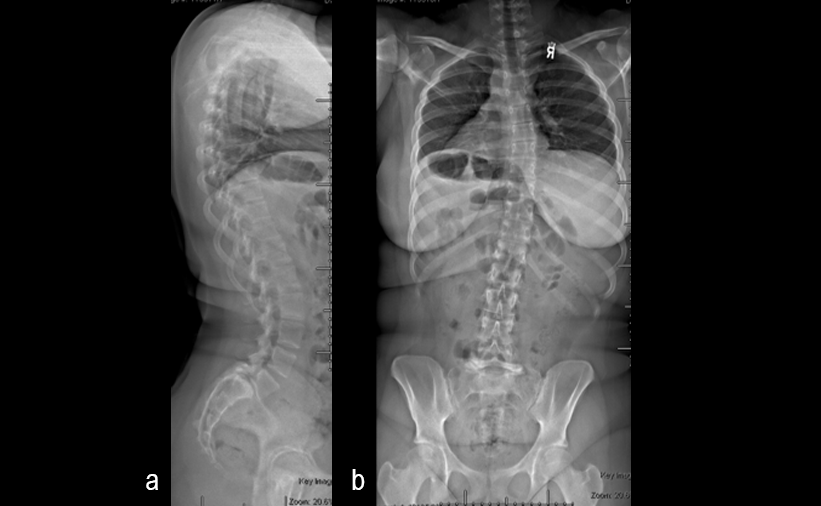
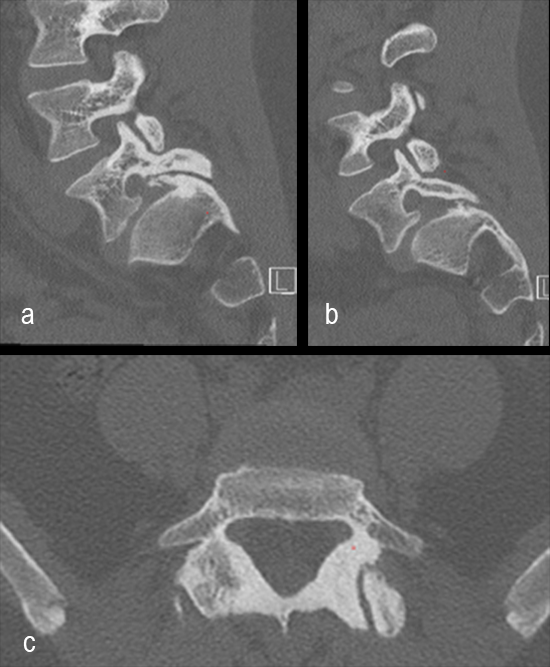
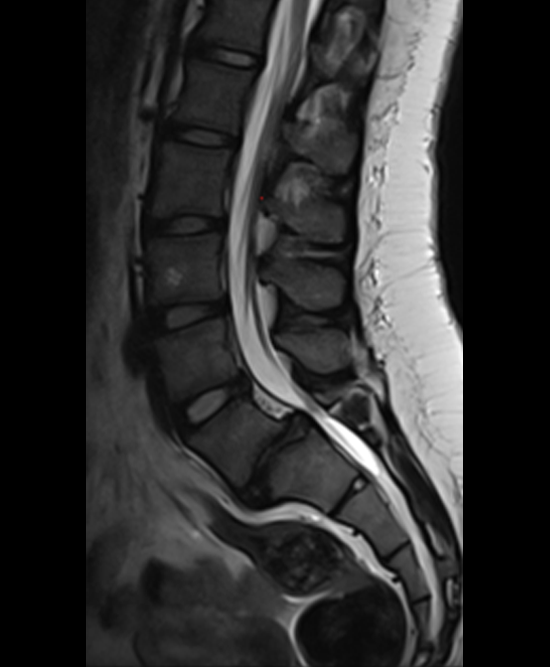
A 17-year-old girl presented to the spine clinic with significant low back pain. X-ray, CT and MRI imaging (Figs 1-3) showed evidence of spondylolisthesis. Upon clinical examination of spinal alignment, the patient was found to have a pelvic incidence of 78°, a lumbar lordosis of 54°, and a pelvic tilt of 51°.
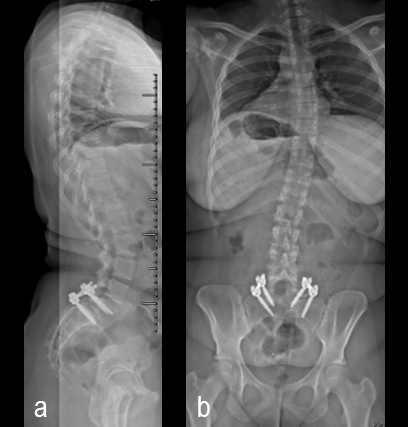
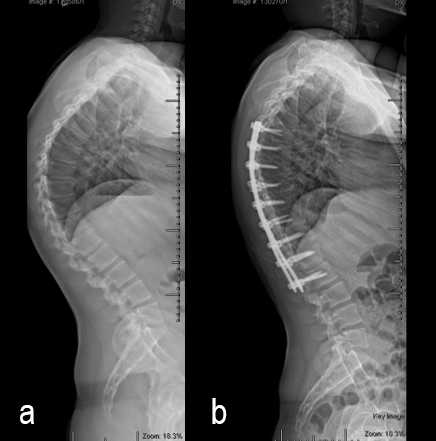
Posterior spinal instrumentation and fusion (PSIF) was undertaken with an interbody fusion at L5-S1. L5 laminectomy and sacral dome osteotomy followed by pedicle screw instrumentation using TriALTIS™ implants helped reduce the lumbosacral kyphosis and translation of L5-S1 and promote fusion across this level (Fig 4a-b).
Case 2
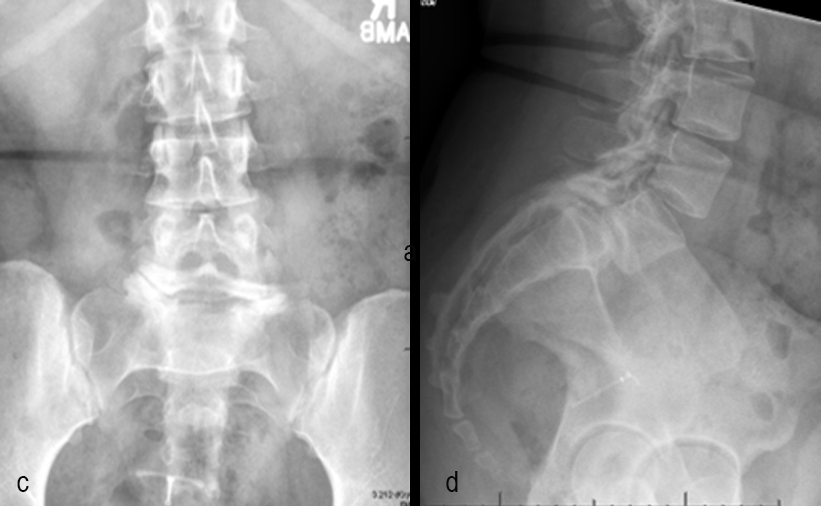
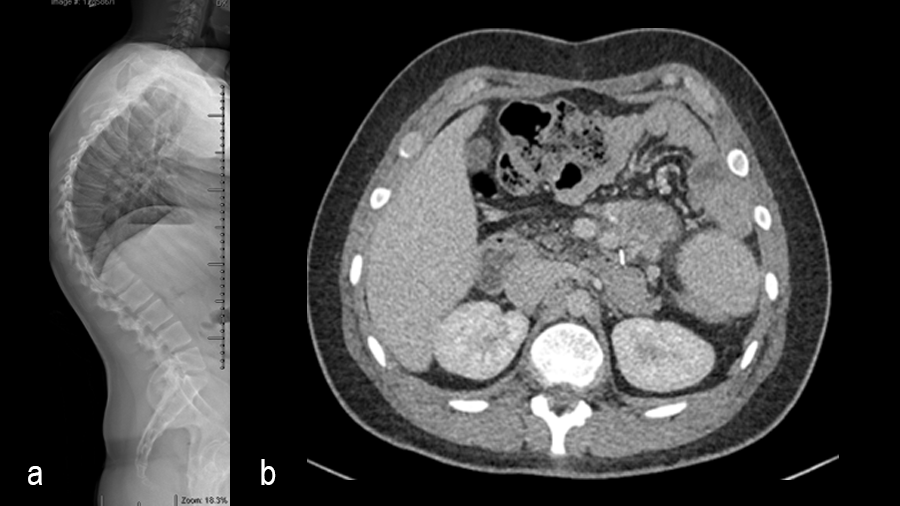
A 19-year-old man with colon cancer was admitted to the spine clinic with pain and progressive kyphosis (Fig 5a). Preoperative CT imaging (Fig 5b) showed a residual mass abutting the superior mesenteric artery.
Surgery was undertaken with TriALTIS™ posterior spinal instrumentation system from T5 – L1 to correct and support the kyphotic deformity (Fig 6a-b). The upper and lower instrumented vertebrae were selected to allow symmetry of fixation above and below the apex of the kyphotic curve. Care was taken not to overcorrect the hyperkyphosis given the abdominal mass abutting the superior mesenteric artery. During surgery, multiple sequential reducers were used to achieve cantilever reduction of the deformity (Fig 7).
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.