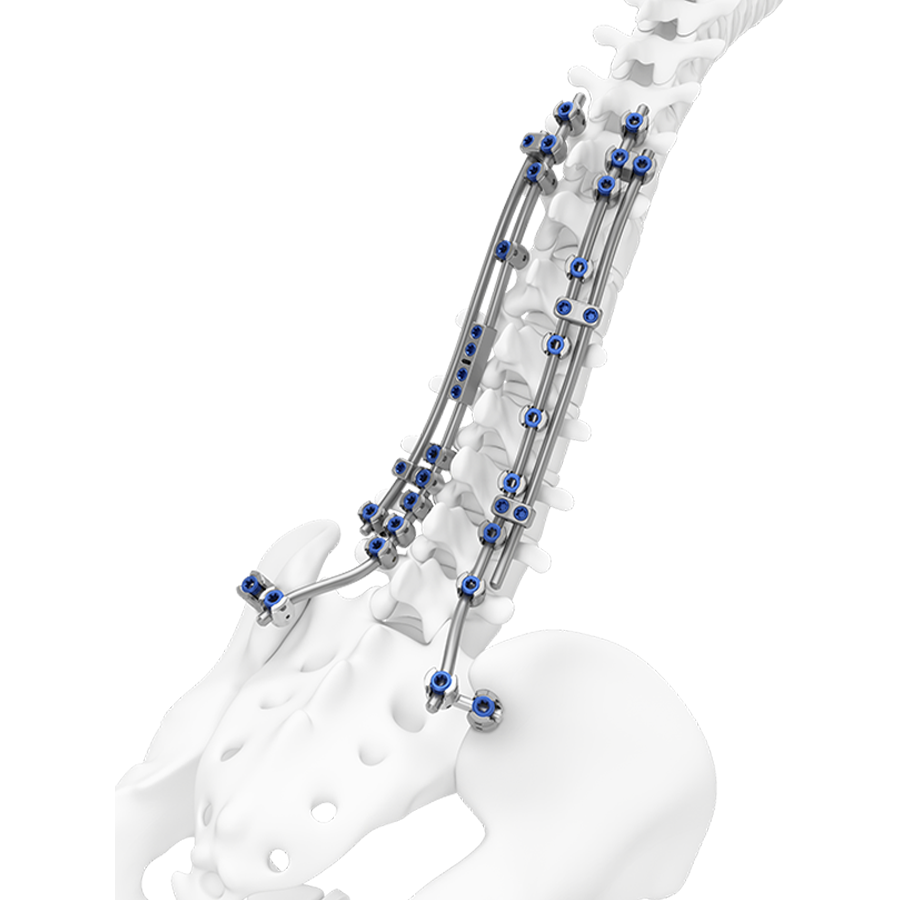
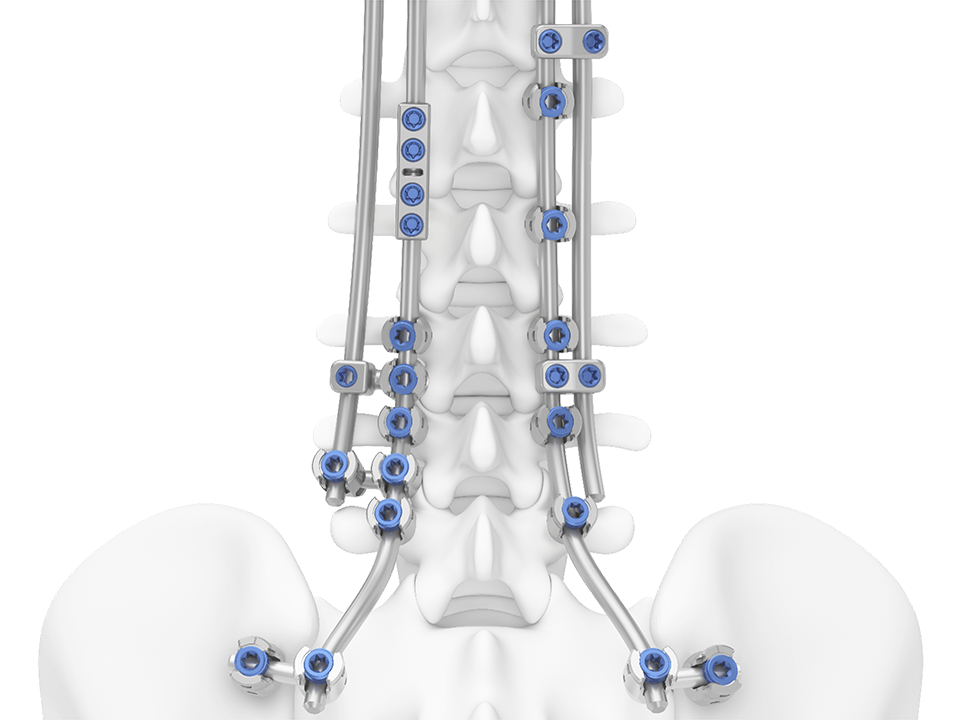
TriALTIS™ Revision System
Tom Fischer, Sudhir Warrier, Alex Lluch, Martin Langer, Stephen Tham
The TriALTIS™ Revision System (Fig 1 and Fig 4) is a new module of rod connectors and surgical instruments, designed as a complementary extension to the original TriALTIS™ posterior thoracolumbar spine implant system. The new components broaden the TriALTIS™ platform’s array of implant and instrument options, enabling tailored solutions for complex thoracolumbar spine revision cases across diverse patient populations.
Connectors
The new connectors (Fig 2) are designed to accommodate different rod configurations including parallel (side-by-side), axial (end-to-end), and lateral. Several different rod capture mechanisms are available, including top loading, side loading, and closed. The system also offers articulating and multiple centerline offset connectors, providing intraoperative flexibility. The connectors feature an integrated set screw, allowing the surgeon to connect to a rod or a connector holder instrument.
- The TriALTIS™ connectors are compatible with other systems as follows:
- 5.5 mm rods: Expedium 5.5 mm hooks and connectors
- 5.5 and 6.0 mm rods: Expedium 5.5–6.35 mm variable connectors
- Matrix Transconnector
- Symphony
Connector instruments offered within the new system allow efficient insertion and manipulation and also provide counter-torque capability for efficient connector handling. Fig 2 shows the unilateral inserter and the connector holder.
Revision Screws
The TriALTIS™ Spine System includes a specialty revision screw offering, with screw diameters ranging from 7.5–12.0 mm. Using larger diameter screws in revision surgery may decrease the likelihood of postoperative complications such as screw pullout [1] in patients requiring revision surgery to the lumbar spine.
Design features
The large diameter favored angle revision screws (Fig 3) are compatible with both 5.5 mm and 6.0 mm rods and have 46° of motion in the favored angle design. The screws feature a quick catch tip for immediate engagement with bone and ease of insertion into the pedicle [2]. The thread design allows a 6 mm screw advancement with one complete screwdriver turn.
The specialty revision screw shank offering includes:
-
Solid Cortical Fix (available in lengths of 30–60 mm)
-
Cannulated Fenestrated Cortical Fix screws (available in lengths of 30–60 mm)
-
Solid Dual Lead (65–110 mm)
-
Solid Partial Thread (65–110 mm)
-
Cannulated Partial Thread (65–110 mm)
Instrumentation
The TriALTIS™ System Revision Screw portfolio is offered in combination with dedicated instrumentation. The revision screw offering shares standard features with all TriALTIS™ Screws, enabling consistent instrumentation across the platform.
- TOP NOTCH™ feature
- Tapered head design
- Rocker connection
- T27 drive feature
Indications (US)
The TriALTIS™ Spine System is intended to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion in the treatment of acute and chronic instabilities or deformities of the thoracic, lumbar and sacral spine.
Full US Indications in IFU – 0902-90-407
Indications (EU)
The TriALTIS™ Spine System is indicated for the following in skeletally mature patients:
- Degenerative spine disease
- Deformities/Instabilities
- Tumors
- Trauma
Full oUS Indications in IFU – 0902-90-432
References
- Varghese V, Venkatesh K, Gurunathan SK. Comparison of pullout strength of pedicle screws following revision using larger diameter screws. (2019) Medical Engineering & Physics, 74: 180 –185.
- DePuy Synthes. TriALTIS™ Evidence Generation Engineering Analysis: Quick Catch and Tapered Self‐Tapping Tip Design. May 9, 2023. Windchill Document No. 501380612 Rev. A
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.