CMF Distractor
Scott P. Bartlett, Richard A. Hopper, Satyesh Parmar, Majeed Rana, Alberto A. Rocha Pereira
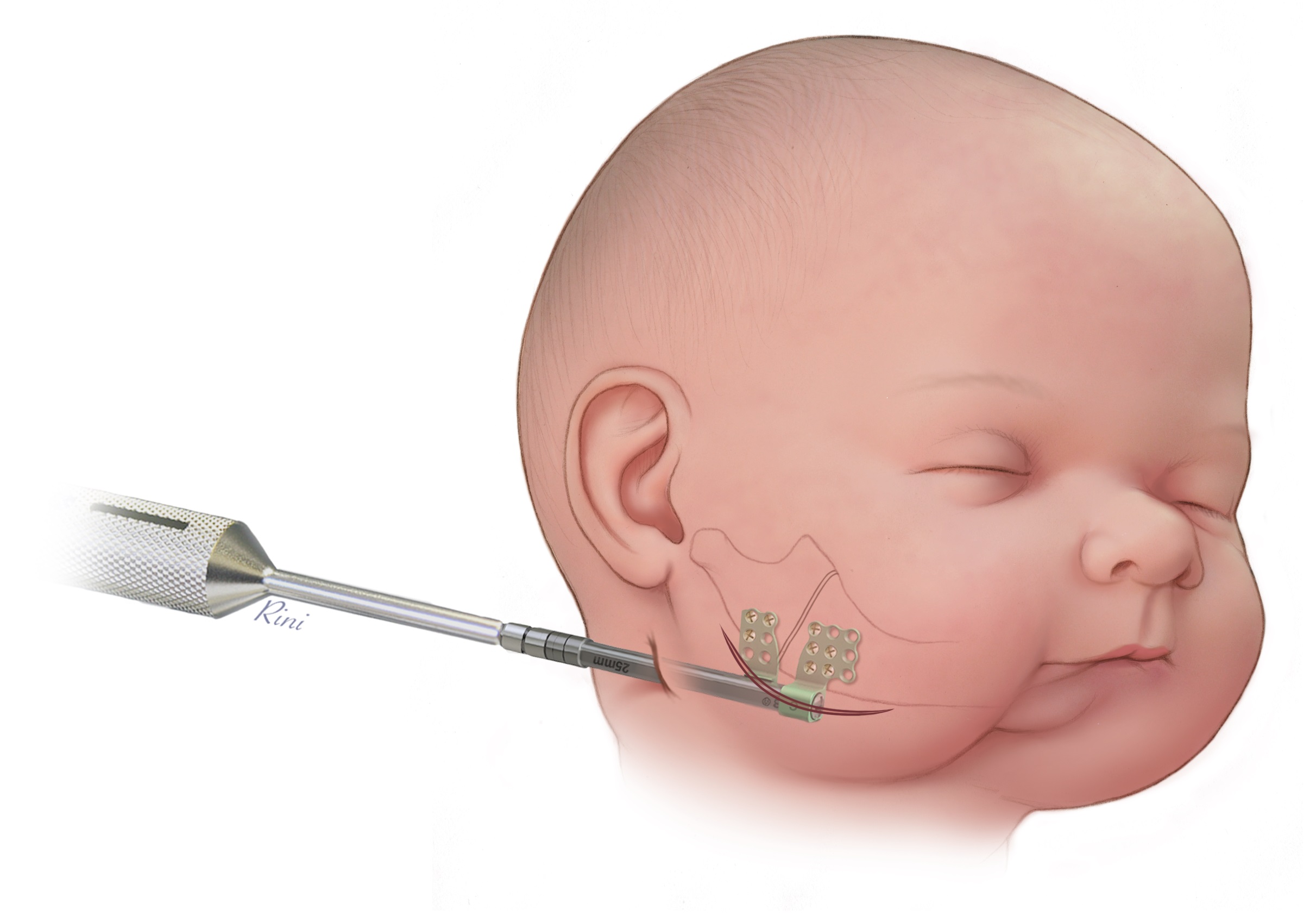
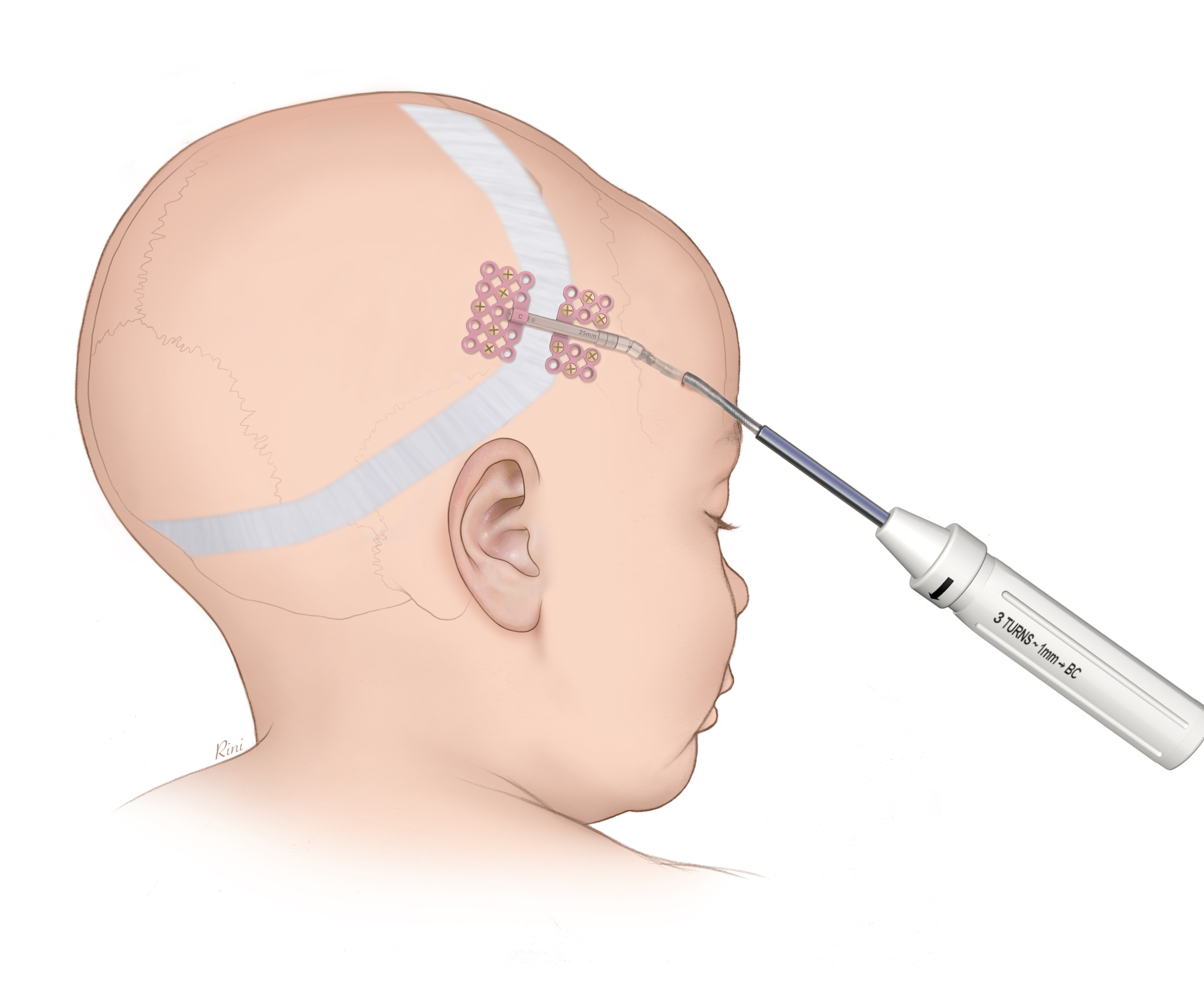
CMF distraction is used for lengthening or bone transport to correct congenital deficiencies or acquired (posttraumatic, post-surgical or post-infectious) defects in the mandible (Fig 1) or the cranium (Fig 2). Gradual expansion allows for new bone formation and soft-tissue adaption both in pediatric and adult patients. With existing systems three main issues have raised concerns. The possibility of wrong-way activation of the system, the inadvertent movement by the system and difficult screw engagement and removal.
The new CMF Distractor is visually similar to the old system (Craniofacial Distraction System), except that the distraction bodies are approximatively 5 mm longer.
New ratcheting mechanism
The new CMF Distractor has a spring clip (detent tab) to avoid inadvertent reversed movement of the distractor. In addition, the new Patient Activation Instrument contains a one-way ratchet clicking when turned in the wrong direction. With these new features the distractor movement is well controlled and if used correctly will produce reliable results.
Raised Head Screws
In addition to the regular Plus-Drive Screws provided with the CMF Distractor, new Raised Head Screws are available for easier removal. These Raised Head Screws incorporate both a Hex-Drive and a Plus-Drive in the same screw head, with the Hex-Drive screwdriver having a tapered fit allowing for the same retention force as in the Plus-Drive system.
Planning and Performing Maxillo-Mandibular Distraction Osteogenesis
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.