Locking Compression Plate 1.5 System
Brian Beale, Randy J Boudrieau, Michael Kowaleski, Rico Vannini
When it comes to fracture treatment in felines and small breed canines, a variety of plating solutions are needed. For example, fractures of the radius and ulna are very common in small breed dogs, yet besides the small size of the bones, it is also known that there can be a decrease in vascular density to the distal radius in these dogs compared with normal sized dogs.
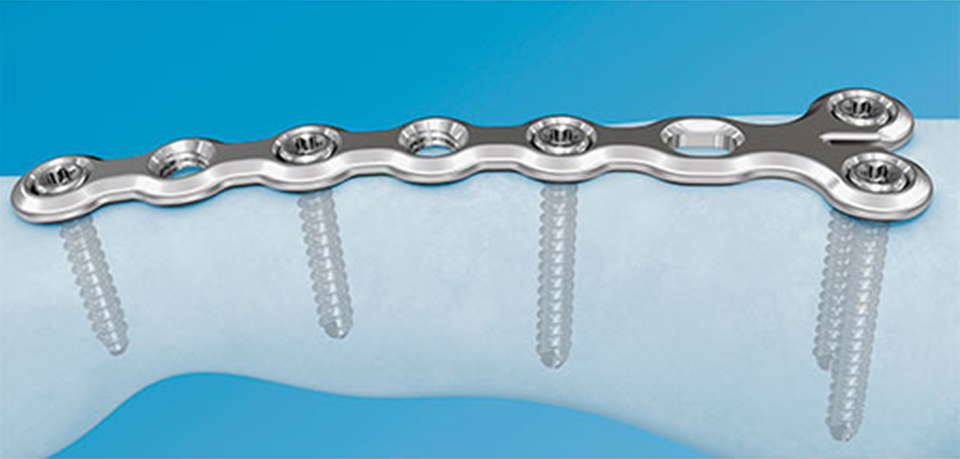
To address these issues, the veterinary plating portfolio has been expanded to include the Locking Compression Plate (LCP) 1.5 system (Fig 1). 1.3 mm locking and cortex screws are compatible with 1.5 mm LCP plates.
This system, which includes straight, T shaped, notched head T, and H shaped plates, has been available for human hand surgery for several years (Fig 2). Because the long-bone geometry of cats as well as toy and miniature breed dogs is similar to the long-bones of the human hand, the contours of the plates fit these animals well. The thickness of the plate is also well-suited for these patients to reduce the chance of stress protection and bone resorption, which is a common problem in toy breed dogs.
The LCP 1.5 system is low profile with rounded edges to minimize soft tissue irritation. To apply this system at different implantation sites, the plates can easily be bent or cut with the respective instrumentation. Combination holes in the straight plates allow the use of cortex or locking screws. Thus, the fracture site can be compressed by placing the screw in the dynamic compression unit portion of the hole to achieve increased fracture stability. Two possible indications for the LCP 1.5 system are distal radius fractures in toy or miniature breed dogs and cats and hyperextension from rheumatoid arthritis.
The adaptation of the LCP system from the human hand to cats and miniature and toy breed dogs addresses a clinical need and provides a perfect solution for many long-bone fracture fixations in small dogs and cats as well for the stabilization of flat-bone fractures such as pelvic or scapular fractures in these animals.
Case 1: Pancarpal arthrodesis
(Case provided by Antonio Pozzi, Florida, USA)
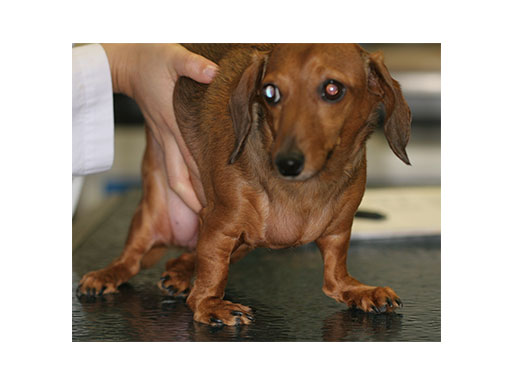
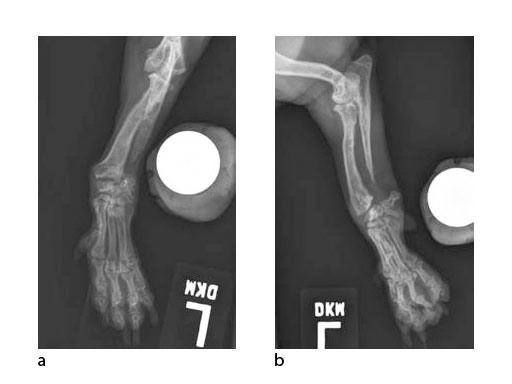
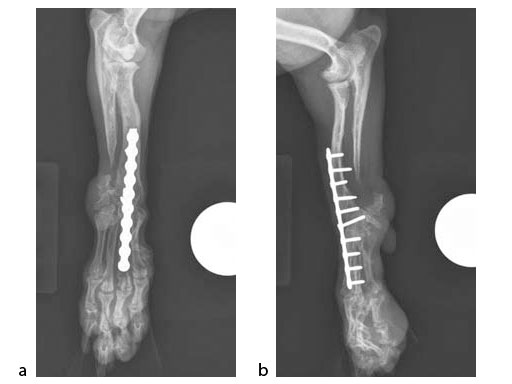
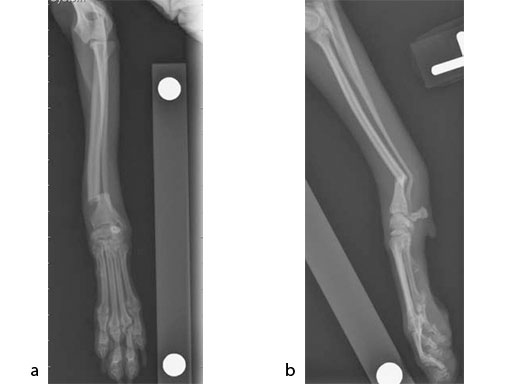
A 5-year-old female 3.5 kg dachshund with bilateral carpal hyperextension secondary to erosive polyarthroplathy (rheumatoid arthritis) (Fig 3, Fig 4) was treated bilaterally with a staged pancarpal arthrodesis with straight LCPs. The right carpus was operated first and healed in 10 weeks. The left carpus was operated 5 months after the initial surgery (Fig 5). Complete healing was noted 3 months postoperatively (Fig 6).
At the most recent follow-up 20 months postoperatively (first arthrodesis), the dog was comfortable and very active. The plates were selected based on the small bone size and the risk of secondary osteopenia. Locking screws are important to prevent screw loosening in patients with poor bone quality, especially in the case of pancarpal arthrodesis where high bending forces must be neutralized.
Case 2: Open reduction and internal fixation of distal radius and ulna fractures
(Case provided by Amy S Kapatkin, California, USA)
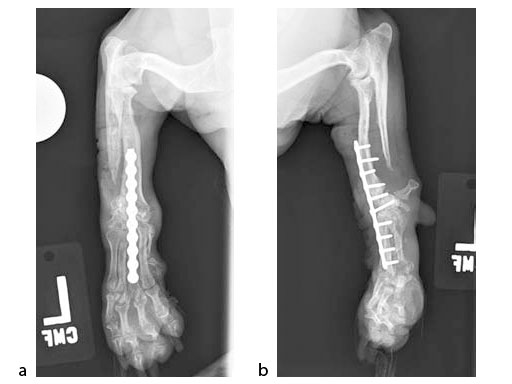
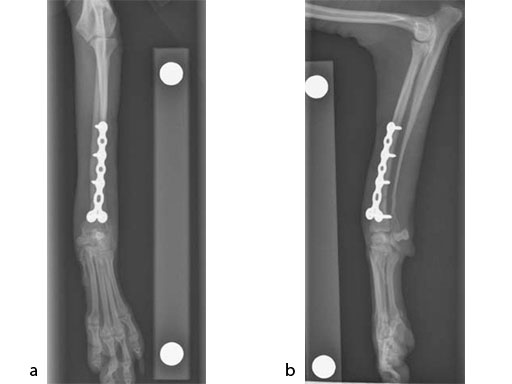
The patient was an approximately 2-year-old 1.5 kg miniature poodle with a radius and ulna fracture of the left thoracic limb. Although there are (1.5 mm screw) cuttable plates available for these small breeds, they require disruption of the periosteum and sometimes the distal fragment has room for only one screw if a straight plate is used. The tiny T plates, using cortex screws, are often too short; ideally at least 3550% of the limb length has to be spanned. In traditional plates, every hole must be filled and contributes to stress protection of the bone. These plate options also do not permit the use of locking screws. Therefore, the condylar LCP 1.5 was a perfect plate for these fractures (Fig 7).
An open but do not touch approach was performed with 3 proximal screws and 2 distal locking screws inserted in the LCP (Fig 8).
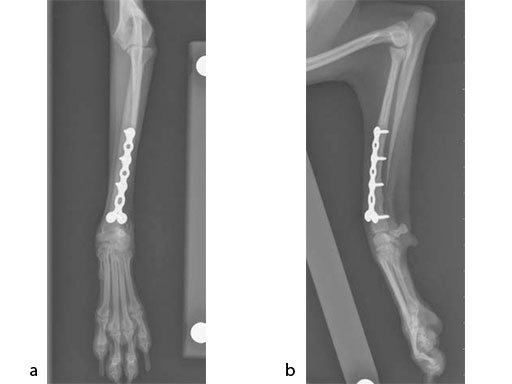
After 3 months, the previous fracture of the radius was no longer visible and there was smooth callus in the region of the previous fracture (Fig 9). There was bone loss at the distal ulna, most likely due to stress shielding. Plate removal can be considered at 6 months if bone resorption is a concern.
Small Bones, Small Plates: Clinical Application of Mini LCP
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.