Veterinary Screw Targeting Clamp (STC)
Fabrice Rossignol, Janik Gasiorowski, Kati Glass, Christopher Lischer, Michael Kowaleski, Jeffrey Watkins
A targeted screw placement system permits improved accuracy of screw insertion when used in conjunction with intraoperative imaging. Improved accuracy of screw insertion gives more consistent, confident fracture repair, particularly through minimal invasive approaches and in challenging anatomical locations, such as distal phalanx and navicular bone fractures. It is also a safe and accurate placement of specific screws into subchondral bone cysts close to joint space.
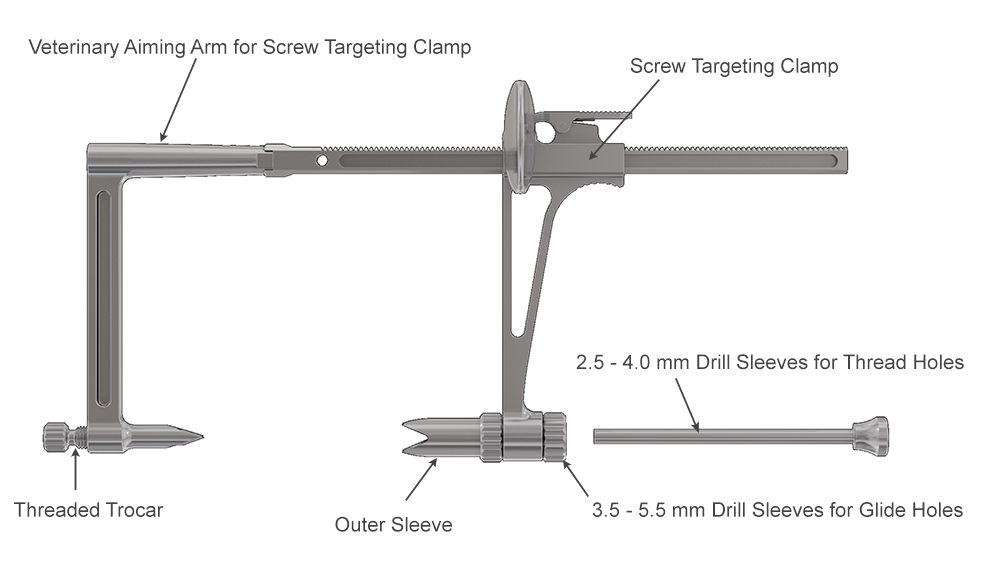
The Screw Targeting Clamp (STC) developed by the AO Technical Commission's Foot and Ankle Expert Group has many useful features for veterinary use. However, available drill guides for the STC do not accommodate the screw sizes frequently used in large animal surgery. Therefore, large animal veterinary-specific sleeves were created for the existing STC to allow placement of 3.5−5.5 mm screws entirely through the STC maintaining alignment.
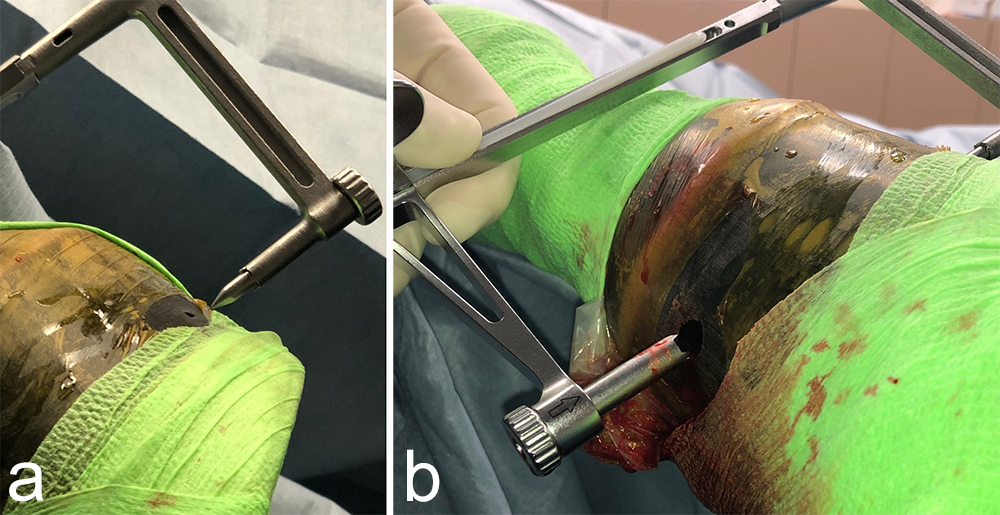
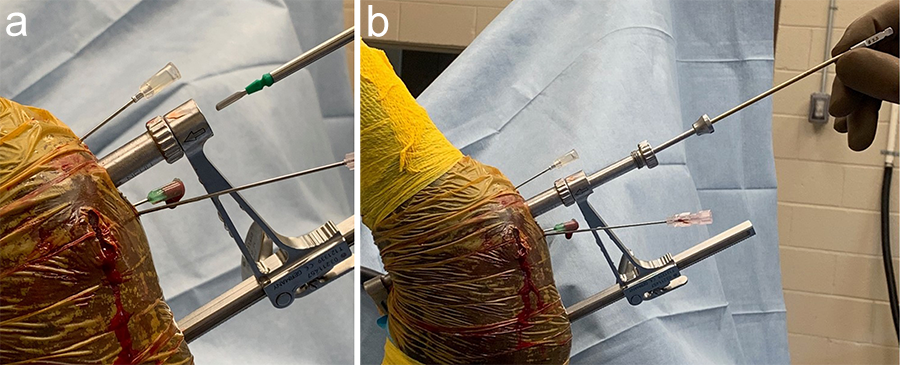
The Foot and Ankle Screw Targeting Clamp was designed to provide the ability to maintain compression and targeted screw insertion. When evaluated in equine bone, clamp bending was observed on application of the compression function. The aiming arm was modified to improve stiffness for use in equine bone, and sleeves were developed to accommodate the screw sizes used in equine surgery. Angled outer sleeves were designed to secure placement of the STC on oblique bone contour (Fig 1).
With its unique design, the STC will hold a bone fragment in reduction and applies a modest amount of interfragmentary compression to maintain stability of the fragments during the procedure. The STC can be associated with standard bone clamps placed adjacent to it if a stronger compression is needed. It also allows all processes necessary for insertion of a cortex screw in lag fashion to be completed through the clamp, from initial drilling to final screw tightening. Screw sizes supported include 3.5, 4.5, and 5.5 mm screws. An adjustable rail helps to ensure ideal screw placement.
Once the fracture is reduced, the STC is placed using imaging control to verify that the trajectory of drilling and screw placement is precise. With the provided insert sleeves, the screw hole is prepared, and the screw is placed while the STC remains in position to protect the adjacent tissues. The STC aids in retaining interfragmentary reduction throughout the entire process of positioning the cortex screw in lag fashion. Once the fracture is reduced and the clamp confirmed to be in the correct location, screw placement can be completed without further imaging.
Feedback from surgeons who tested the new Veterinary Screw Targeting Clamp stated that a more accurate placement of screws can be achieved, and that the system is more stable than the existing targeting clamp and standard drill guides.
Case 1: Subchondral bone cyst at the distal aspect of the middle phalanx
(Case provided by Fabrice Rossignol and Ariane Campos, Grosbois Equine Clinic, Paris, France)
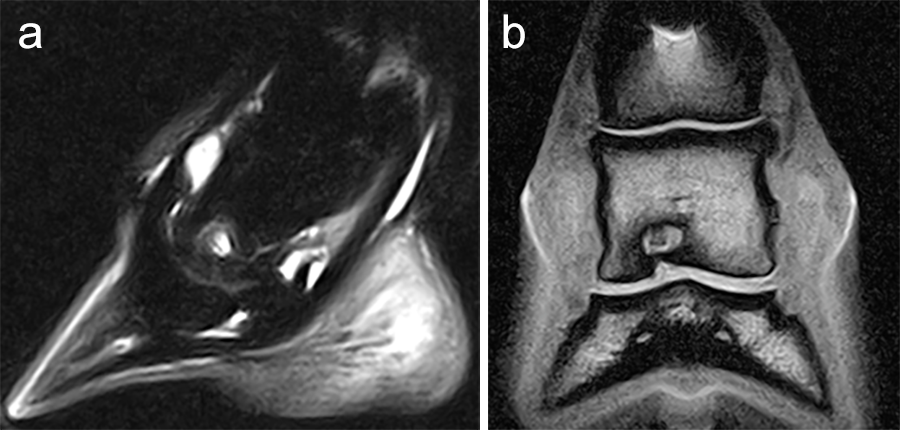
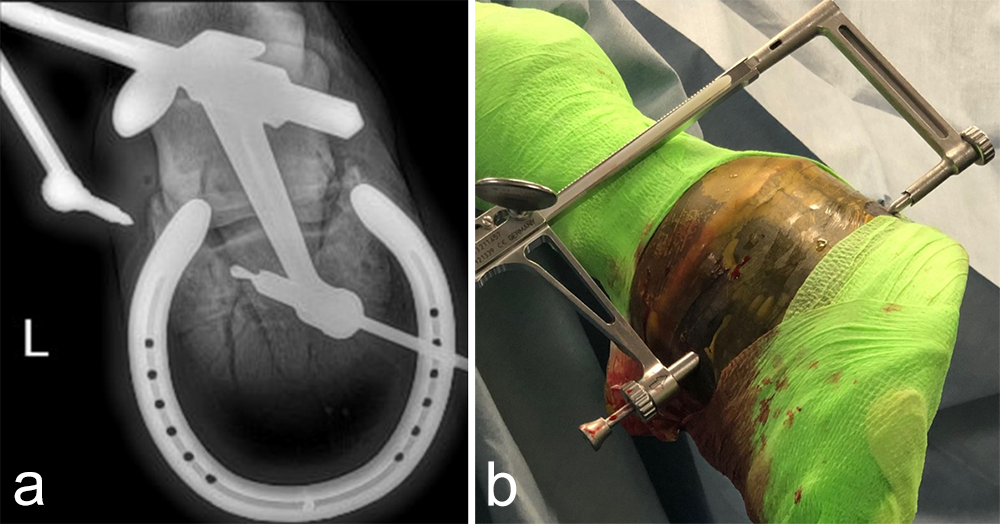
A subchondral middle phalanx bone cyst was identified on a clinically sound 3-year-old Warmblood Gelding during radiographic screening. The osseous lesion was located at the medial and distal aspect of the middle phalanx on the left front limb. Radiographs indicated mild sclerosis of the trabecular bone surrounding the cyst. A magnetic resonance imaging examination confirmed discrete communication with the distal interphalangeal joint (Fig 6). The treatment of choice was surgery, which included placing a cortical lag screw or compression screw across the subchondral bone cyst. This technique aims to promote new bone formation by altering the biomechanical bone environment. A 4.5 mm headless compression screw was placed across the cyst with the veterinary screw targeting clamp.
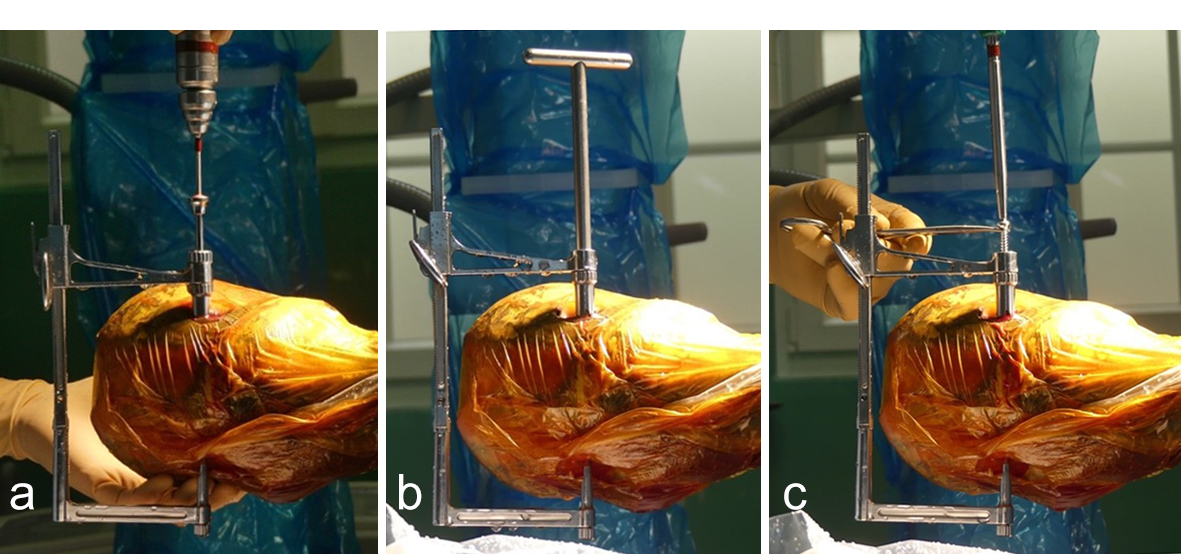
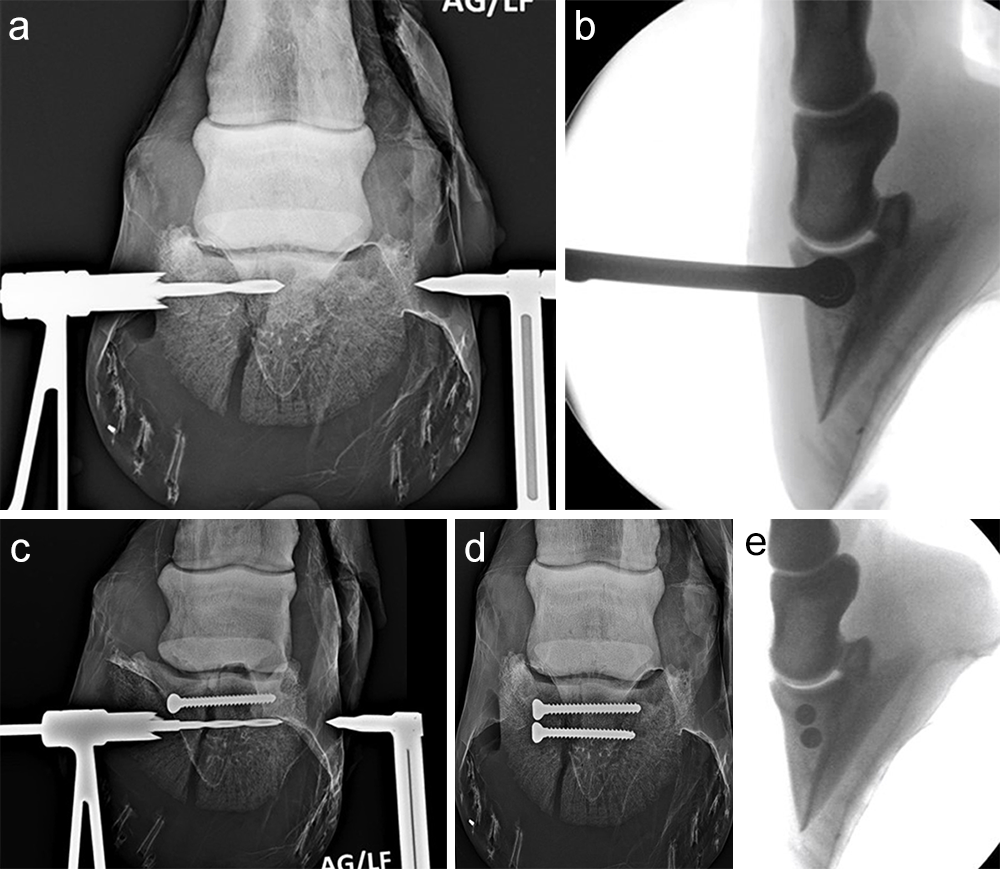
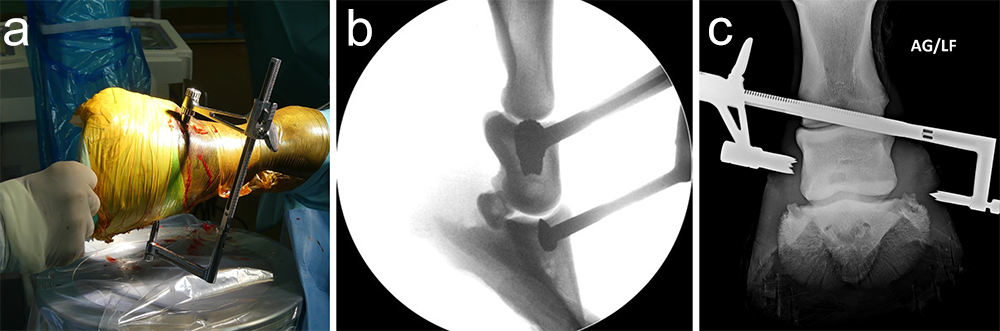
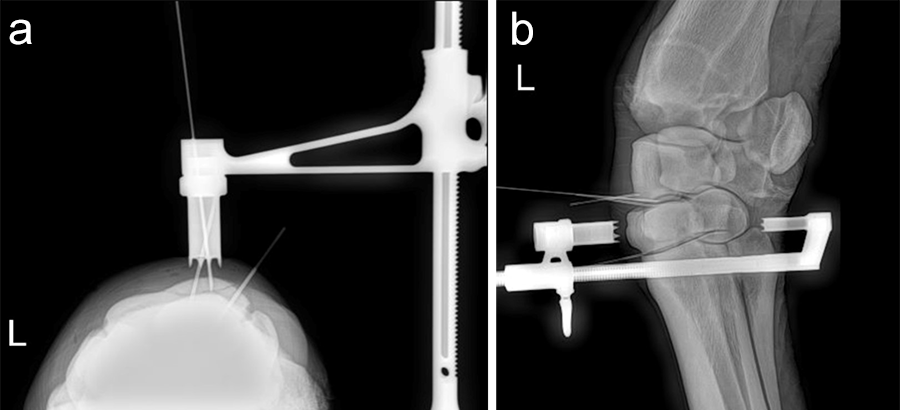
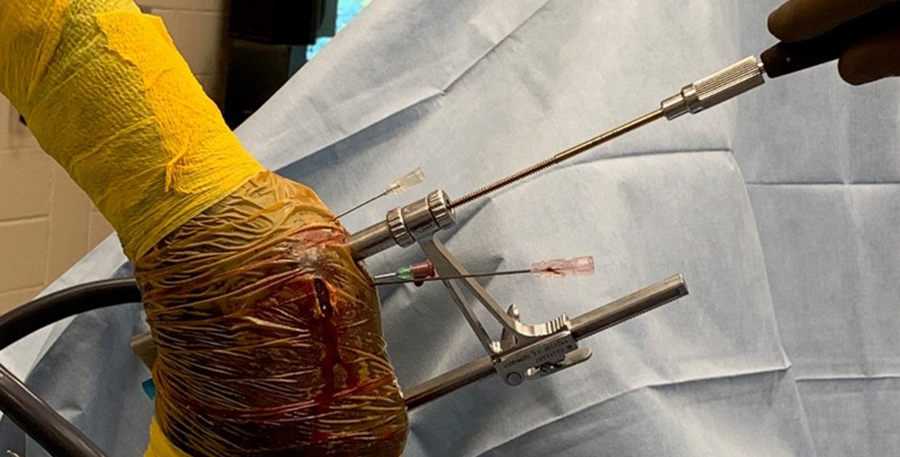
The horse was routinely anesthetized and positioned in left lateral recumbency to be able to access the medial aspect of the left forelimb. The lower limb was aseptically prepared and draped appropriately to isolate the solar region of the hoof from the surgical area, and to allow visualization of the coronary band. The targeting clamp was assembled with the straight outer sleeve and the 4.5 mm drill sleeve. It was positioned under radiographic and image intensifier guidance in an oblique direction to avoid drilling through the proximal hoof wall. Once the surgeons were satisfied with the position of the targeting clamp, the location of the trocar was marked on the draping and a stab incision was made at the level of the outer sleeve. The outer sleeve was inserted in the skin incision and the adjustable rail of the targeting clamp was tightened. The 4.5/3.2 mm insert sleeve followed by the 3.2/1.6 mm insert sleeve (provided in the 4.5 mm headless compression screw kit) were placed in the 4.5 mm drill sleeve (Fig 7).
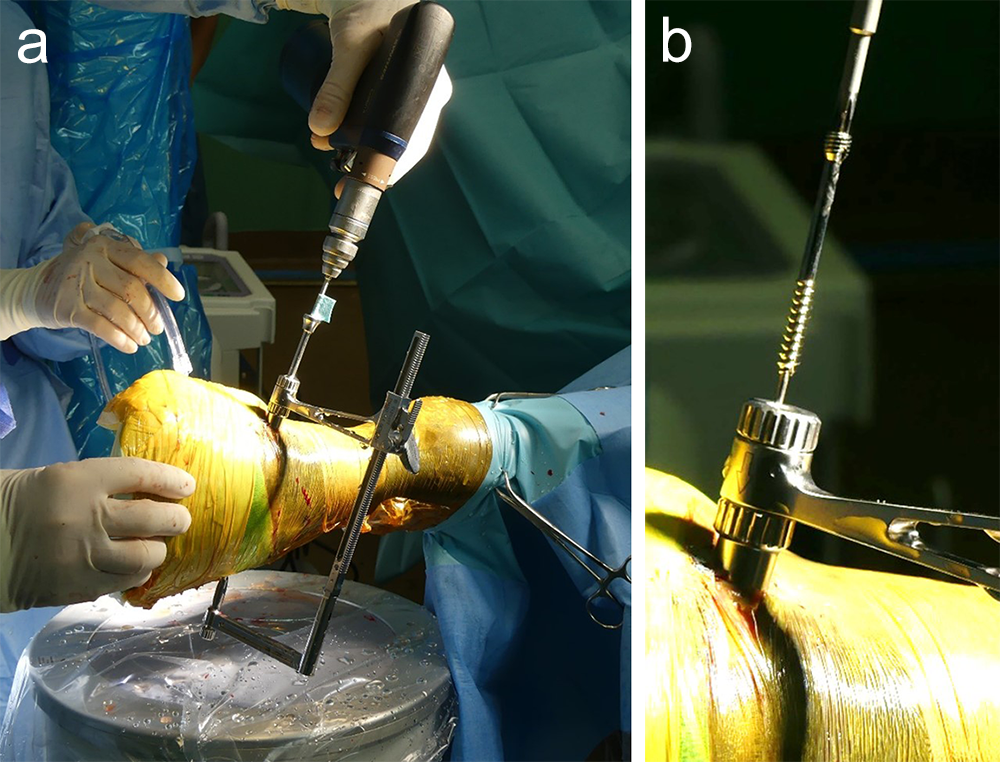
A 1.6 mm guide wire was advanced through the bone to assess the correct positioning of the targeting clamp. The guide wire is centered within the cyst. At this stage, the ideal screw length was measured on the perioperative radiographs. When using these headless cannulated compression screws, remember that the shaft thread length is approximately 40% of the screw length. Ideally, the threaded shaft of the screw is positioned on the trans-side of the cyst. The 3.2/1.6 mm insert sleeve was then removed and the bone was drilled with a cannulated 3.2 mm drill bit to the required length (Fig 8a). Considering the high-bone density at this location, the entire length of the drill hole was tapped by hand with a cannulated 4.5 mm tap through the 4.5 mm drill sleeve. Next, a 4.5 mm headless compression screw was inserted through the 4.5 mm drill sleeve and tightened by hand (Fig 8b). The stab incision was closed routinely. The horse was covered with a two-layer bandage. The total surgery time was 30 minutes.
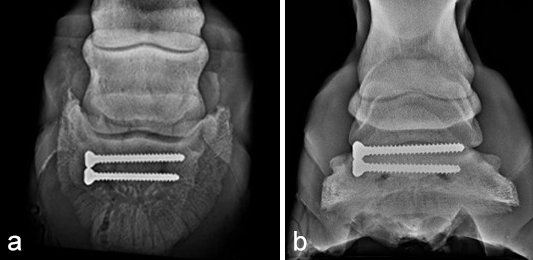
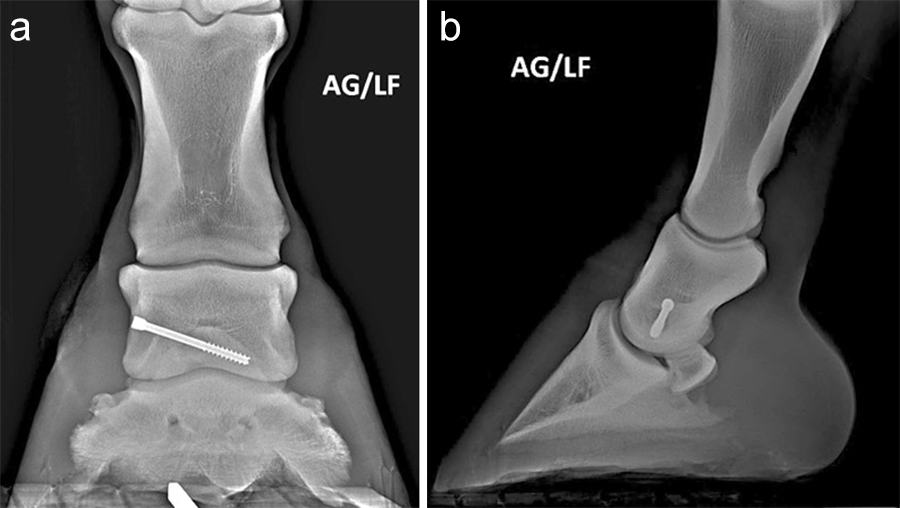
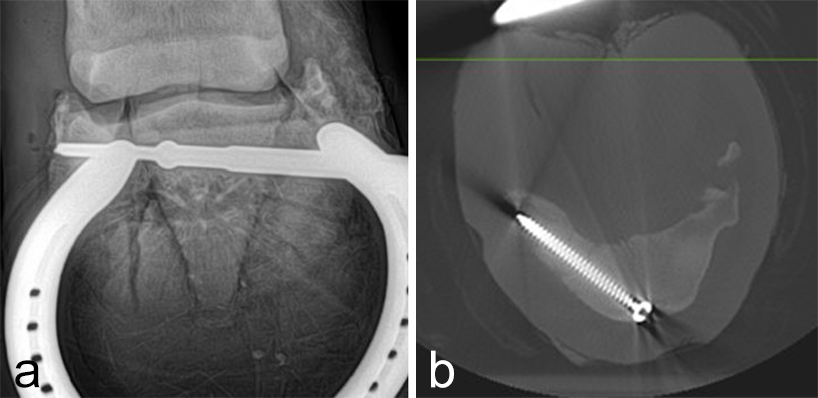
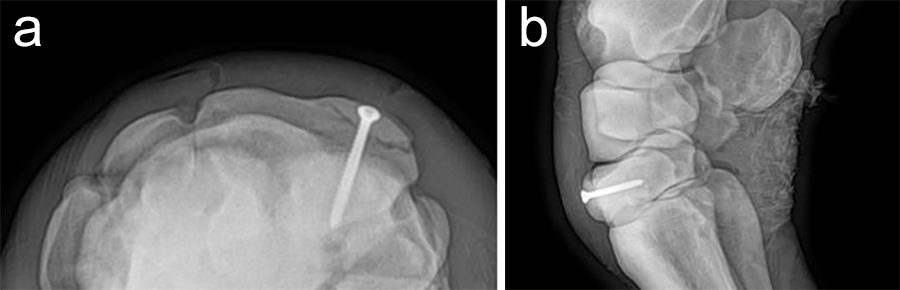
Postoperative radiographs showed accurate screw positioning (Fig 9). The horse was box-rested for 2 weeks and hand-walked for another 6 weeks. Ridden work was resumed after 2 months. The horse remained sound in the postoperative period. Four months after surgery, the radiographic definition of the cyst margins was markedly decreased.
Case 2: Lateral type II fracture of the distal phalanx repair—3-year-old Standardbred racehorse
(Case provided by Janik Gasiorowski, Mid-Atlantic Equine Medical Center, Ringoes, New Jersey, USA)
An articular fracture of the lateral wing of the distal phalanx was diagnosed at the track in this 3-year-old Standardbred racehorse, which was referred for surgery. Internal fixation was planned with a 5.5 mm cortex screw in lag fashion to restore and maintain articular congruency by fracture reduction and interfragmentary compression. Drill trajectory was planned with computed tomographic (CT) guidance with the horse awake and standing. Grids of barium paste dots were placed at proposed entry and projected exit sites. Once the ideal dot was selected from each grid the horse was anesthetized and the hoof prepared for surgery. The veterinary Screw Targeting Clamp (STC) was affixed to the hoof at the ideal dot in the entry and exit grids. Drilling, countersinking, measuring, tapping, and screw placement were achieved through the STC. The CT guidance and the STC allowed placement of the screw between the articular surface and vascular canal containing the terminal arterial arch, and in an orientation that resulted in accurate reduction instead of translation of the fracture fragment and parent bone.
Case 3: Frontal plane third carpal bone slab fracture repair—3-year-old Thoroughbred racehorse
(Case provided by Janik Gasiorowski, Mid-Atlantic Equine Medical Center, Ringoes, New Jersey, USA)
A frontal plane fracture of the third carpal bone was diagnosed at the track in this 3-year-old Thoroughbred racehorse, which was referred for surgery. Internal fixation was planned with a 3.5 mm cortex screw in lag fashion. The Screw Targeting Clamp (STC) placement was guided using needles with radiographic and arthroscopic imaging. From skin incision to screw placement, lag screw fixation was achieved through the STC without movement or removal of the device.
Veterinary Screw Targeting Clamp
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.