Surgical technique guides and design guidelines for 3-D Patient Specific Implants
Richard A. Hopper, Majeed Rana, Rainer Schmelzeisen
New techniques of three-dimensional imaging as well as rapid developments in computer-aided design (CAD) and computer-aided manufacturing (CAM) are changing the face of modern medicine. With the use of digital treatment planning and patient-specific templates, complex surgical procedures can especially be improved; this is true in terms of feasibility and accuracy, and in terms of operating time and clinical outcome. The increasing number of patient specific implant (PSI) options now available in craniomaxillofacial surgery can make treatment planning challenging for surgeons. Although the workflow for timely and accurate planning, production, and delivery of PSI has been well established, the specific steps to use them during surgery is less clearly defined.
The AOTK (CMF), specialized expert groups, and PSI industry partners have identified the need for support for surgeons in treatment planning to improve both clinical reliability and work flow efficiency. They identified an opportunity to clarify the specific surgical steps involved in using this new technology through the creation of detailed technique guides. This will allow surgeons to avoid having to develop a PSI treatment plan from scratch with each new patient. Although technique guides are available for non-PSI implants and devices, due to the custom nature of PSI technology, regulations regarding their design and use remain less established. This can place the onus on the surgeon alone to determine clinical indication, identify the treatment goals, design the device parameters, and incorporate them all into the surgical plan. Although industry staff and engineers are available as a technical resource in PSI planning, the AOTK has identified value in providing surgeons using PSI materials with a stepwise technique guide created by AO clinical experts.
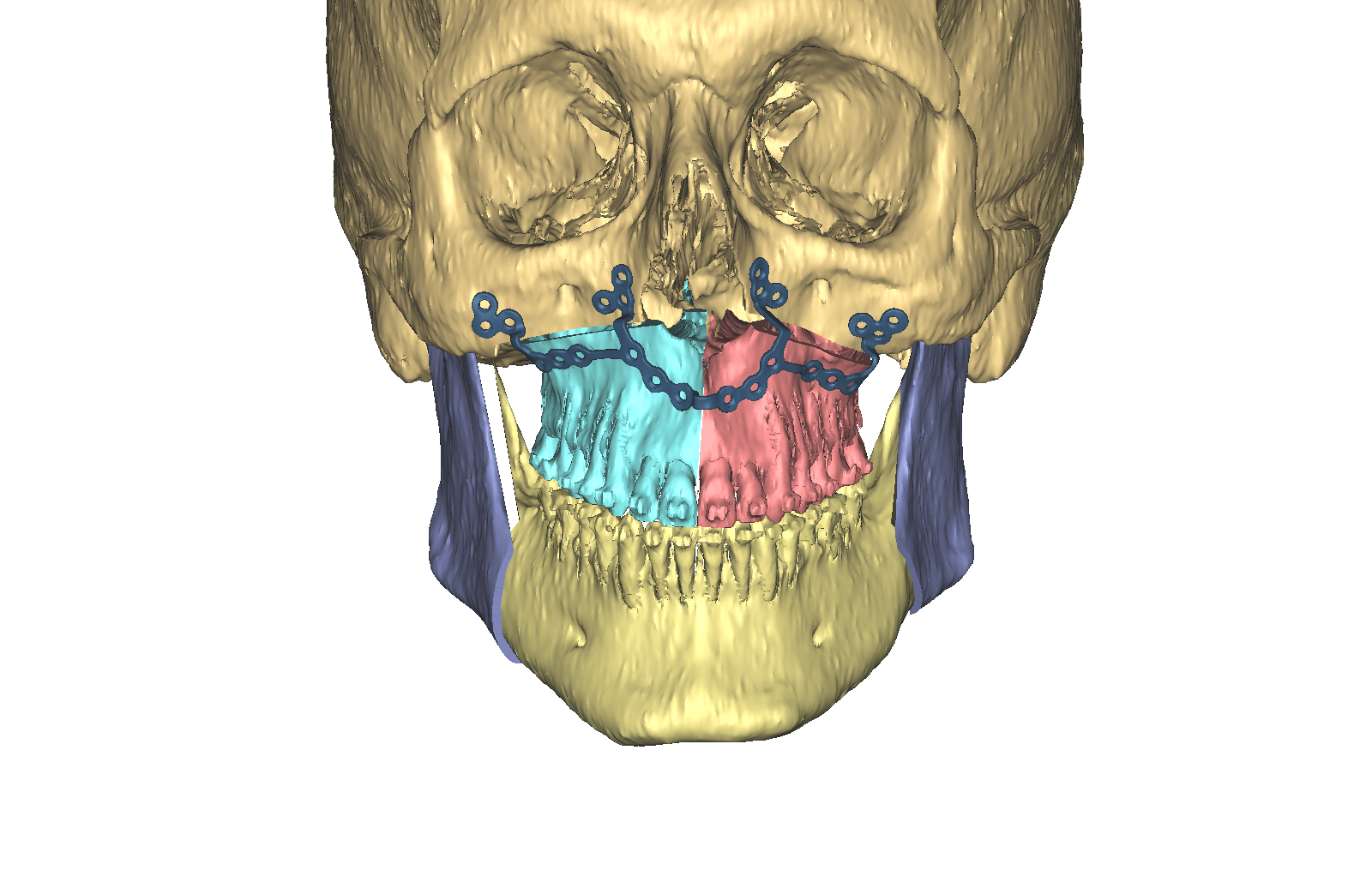
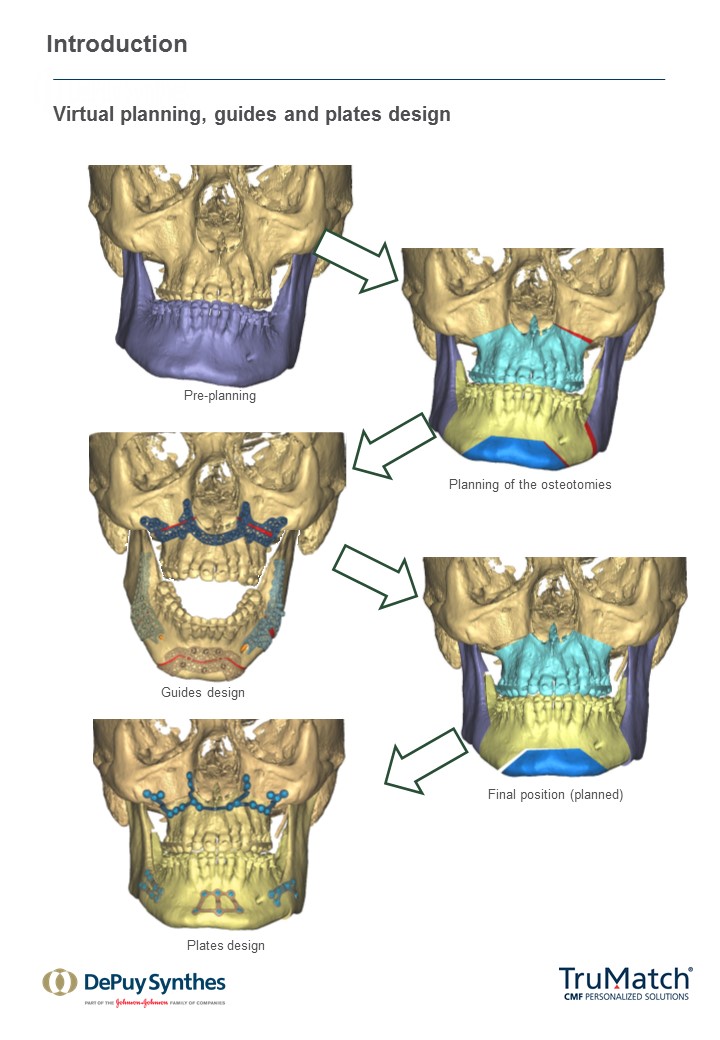
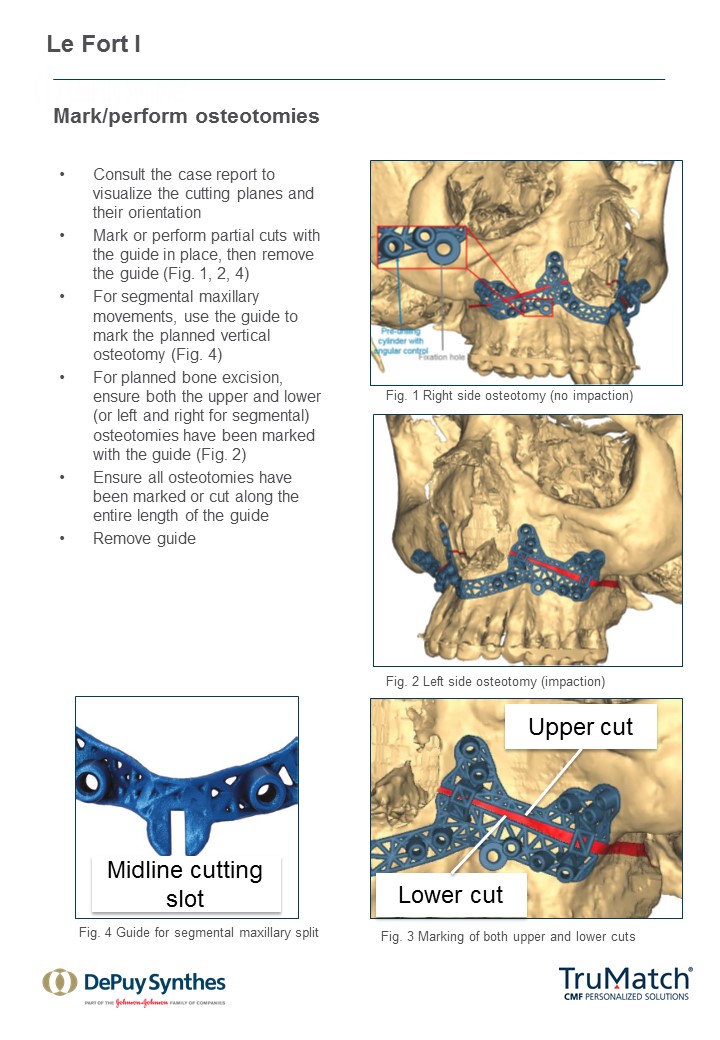
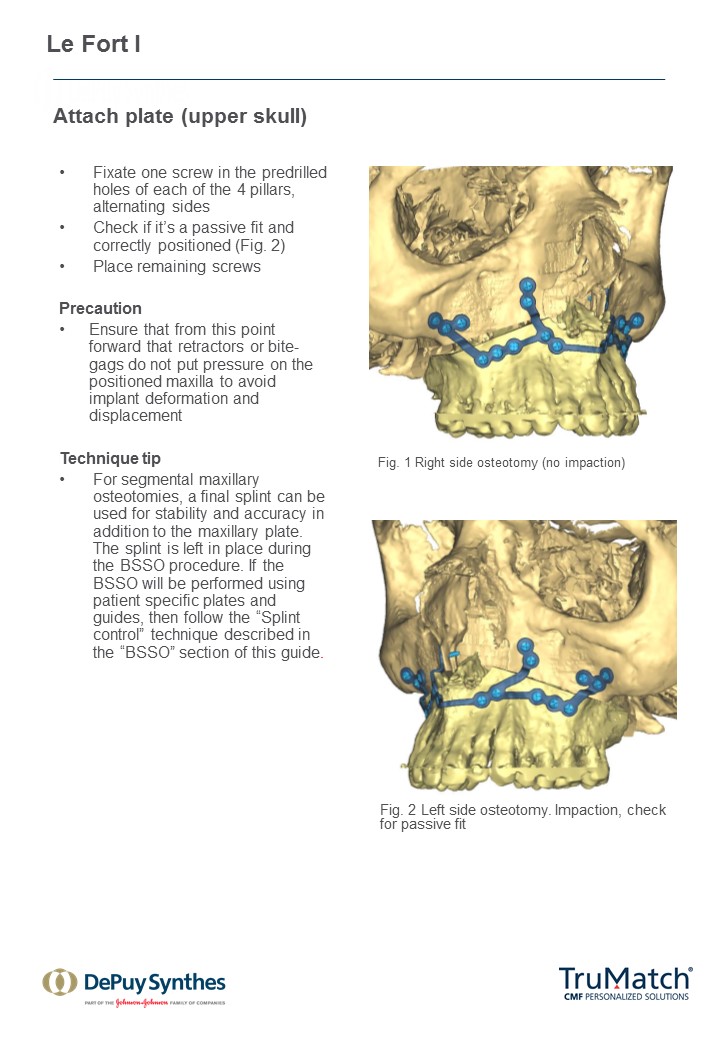
As a first step in this initiative, a technique guide for the TruMatch Orthognathics Platform has been developed with the oversight of the AOTK (CMF), which explains all surgical steps in detail, including insights from experts into how to maximize both safety and accuracy (Figs 15). This project follows the AOTK principle that AO experts involved in the development of a device are also in best position to provide guiding advice on their indications and use.
The potential benefits and actual limits of an integrated 3-D virtual approach for the treatment of patients and requirements for craniomaxillofacial surgery are discussed comprehensively from our experience using 3-D virtual treatment planning clinically. The next steps for this initiative will be to design additional publications that will offer this same guidance during the online designing process of the TruMatch PSI products.
Patient Specific Procedures in CMF: State of the Art
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.