Syncage Evolution System
Paul Heini, Khai Lam
Syncage Evolution represents a new direct anterior or anterolateral interbody cage for the lumbar spine (Fig 1). The anatomical design and wide range of sizes (available in various foot prints, heights, and angulations) facilitate the correction of any anterior interbody problem. Specific and sophisticated instrumentation enables safe and controlled application of the implant.
Primary indications for use include:
Degenerative disc disease
Revision procedures for postdiscectomy syndrome
Pseudoarthrosis or failed fusion
Degenerative spondylolisthesis
Isthmic spondylolisthesis
Anterior column support for osteotomies.
The Syncage Evolution spacer must be applied in combination with supplementary fixation, such as an anterior plate system or pedicle
screws.
Contraindications include:
Vertebral body fractures
Spinal tumours
Osteoporosis
Infection.
Design features
The design of the implant offers increased stability. Its pyramidal teeth provide primary resistance to implant migration, and the large graft volume allows for undercuts and openings in struts to increase graft volume. The middle strut design allows for an improvement to ratio of graft volume to endplate contact, and the diamond shaped anterior and anterolateral interface provides for optimal force distribution from the implant holder to the implant.
Other features include a self-distracting nose, which allows for ease of insertion. The tantalum radiographic marker pins enable visualization of the implant position during insertion.
Material: available in PEEK with 0.8 mm tantalum marker pins.
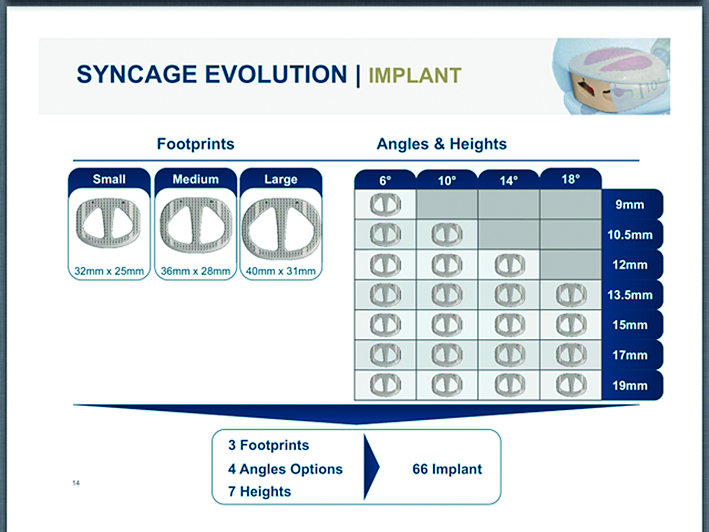
The comprehensive and competitive Syncage Evolution portfolio (Fig 2) boasts an asymmetric anatomical shape for more patient specific implants:
- Footprints: small (32.0 x 25.0 mm), medium (36.0 x 28.0 mm),
large (40.0 x 31.0 mm) - Heights: from 9.0 mm to 19.0 mm (9.0 mm, 10.5 mm, 12.0 mm,
13.5 mm, 15.0 mm, 17.0 mm, 19.0 mm) - Angles: 6 to 18 (6, 10, 14, 18)
- Asymmetric cranial and caudal surfaces with a 3-D convex shape for optimized endplate contact.
The improved instrumentation enhances ease of use compared with other systems in specific surgical phases.
Posterior release tool
The posterior release tool (Fig 3a) is used as an alternative to standard spreaders (Fig 3b).
Features include:
- Allows for progressive and controlled distraction and posterior release
- Broad tips avoid subsidence of the instrument
- Posterior release height is reproducible
- Changeable inserts for mobilization prevent over-distraction.
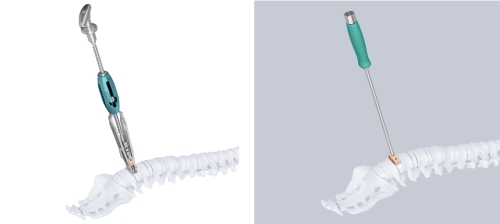
Evolution Squid
The evolution squid (Fig 4a) is used as an alternative to implant holders (Fig 4b):
- Distracts and inserts the implant in one simple step without impaction
- Offers multiple positioning options to recess implant in disc space
- Rails provided for safe implant guidance during insertion
- Thin blades prevent over-distraction during implant insertion.
Evolution trial rasps
Evolution trial rasps (Fig 5) have been specifically designed to help smooth the end-plates and create bleeding to aid with the inter-body fusion.
Case provided by Paul Heini, Bern, Switzerland
Case 1
A 75-year-old female patient presented with postoperative back pain. She had been initially operated on eight years earlier with a laminectomy and fusion from L2 to L4. This proved to be successful for a number of years until a second operation was required for secondary back pain and left side leg pain. An extension of the decompression was performed with stabilization and fusion from L1 to S1. The rationale for this operation was unknown and the surgery failed to improve her symptoms.
The problem to be addressed was the patients back pain and left side leg pain, inclusive of some weakness in her left foot. The pain was present upon weight-bearing, with a pain scale of 9. Her discomfort remained at night. The patient was of slim build and was in good general health. She presented with a limp from her left hip and the dorsiflexion of the left foot was weak (M4).
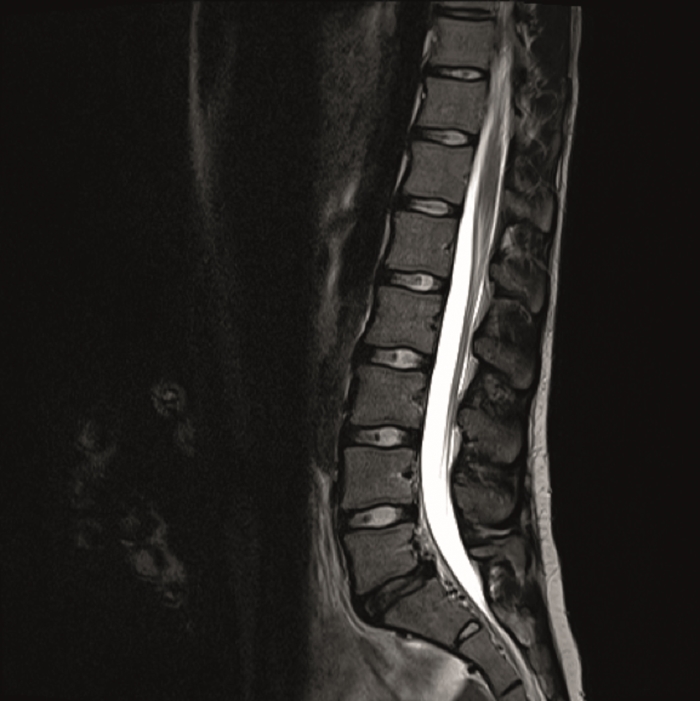
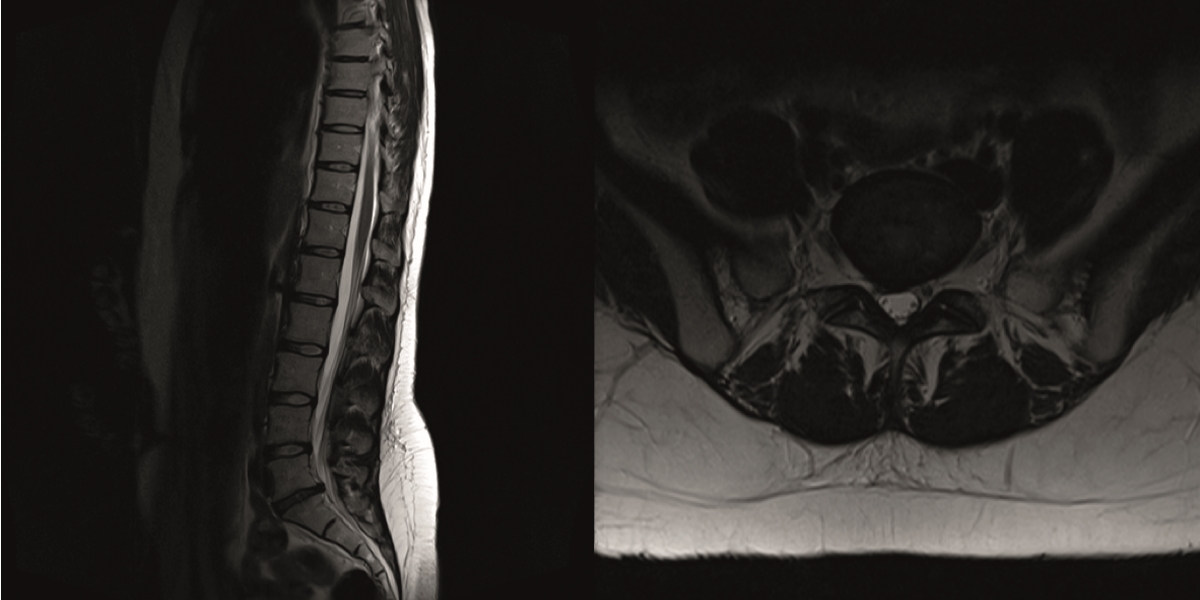
The preoperative standing image of the lumbar spine revealed a flat back with no obvious degeneration of the adjacent segment L1/L2 (Fig 6ab). The implants seemed regularly placed. After wide laminectomy, the spinal canal was open over the whole lumbar spine, illustrated on the MRI scan (Fig 6c).
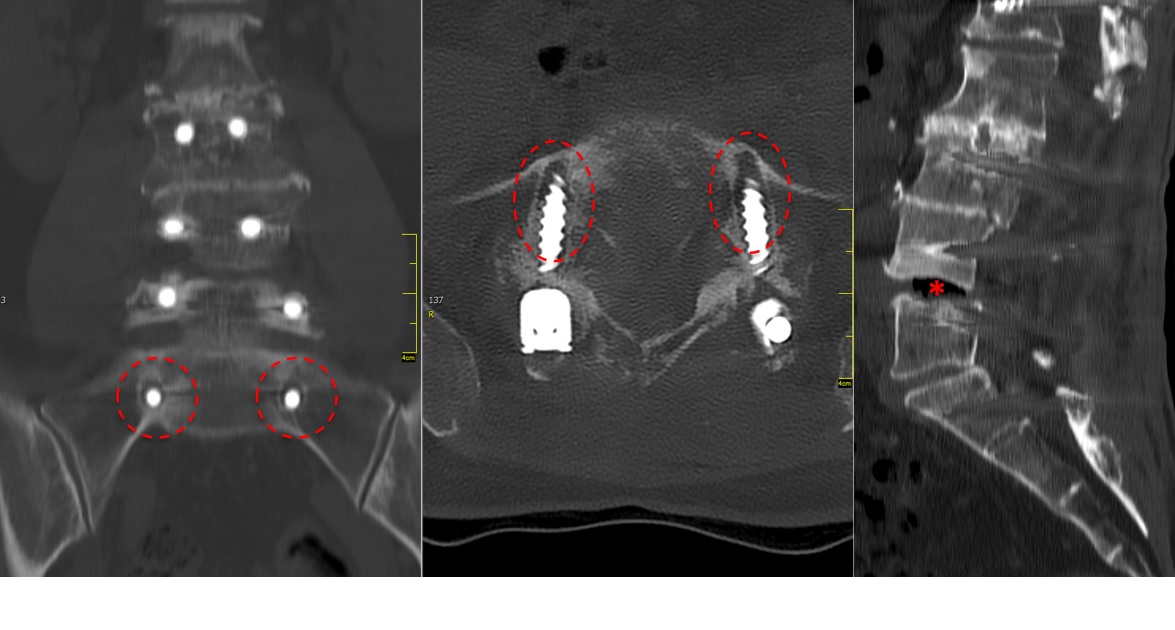
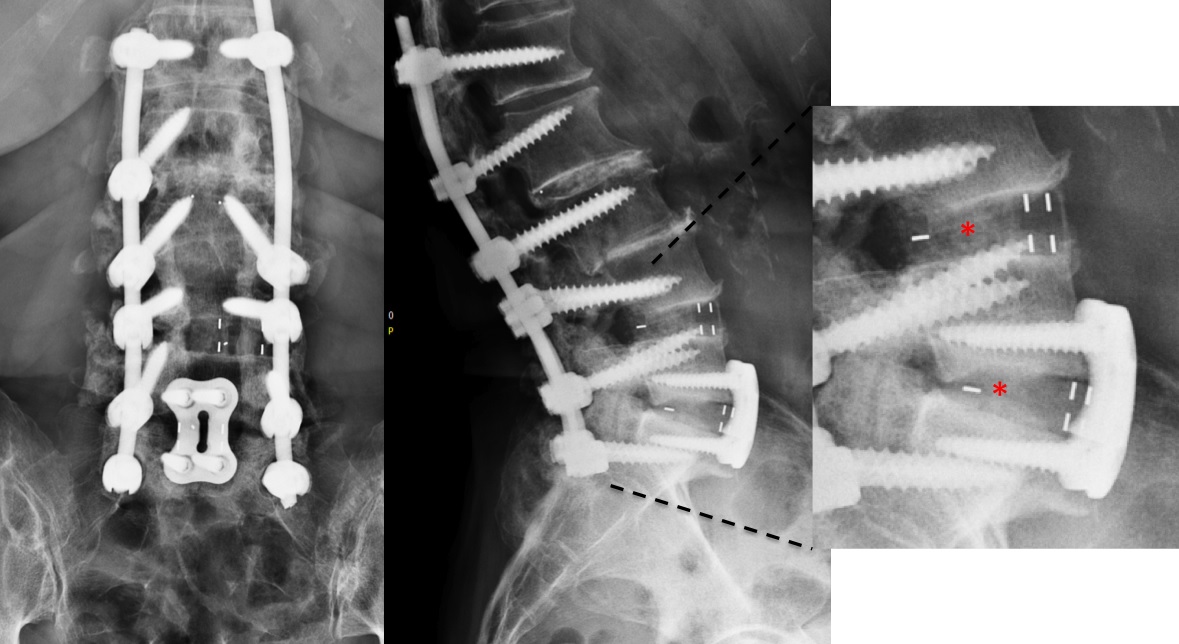
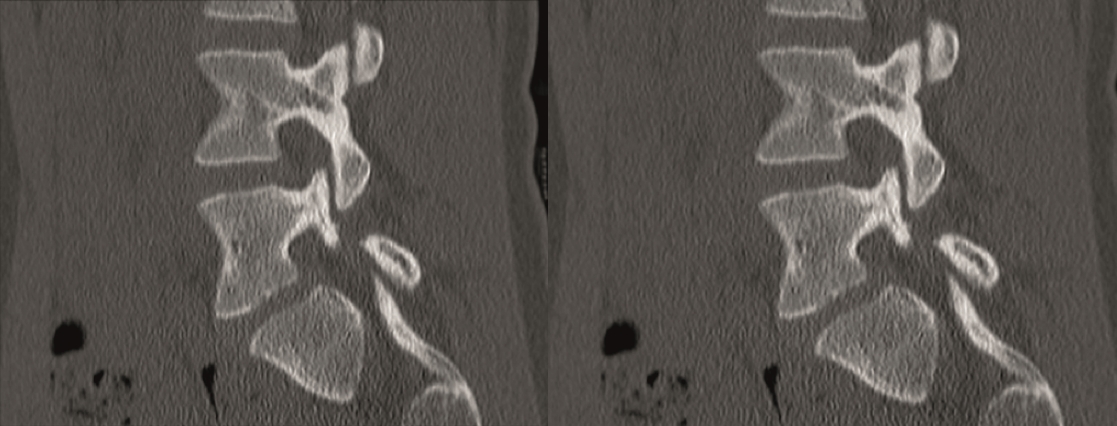
A CT scan allowed a more detailed assessment (Fig 7). There was an obvious nonunion at L5/S1, with loose screws in the sacrum (red circle). Furthermore, there was instability at L4/L5 as the intervertebral disc presented with an important vacuum phenomenon (asterisk). Foraminal stenosis at L5/S1 (not shown) seemed to be the reason behind the persistent leg pain.
The treatment plan was an anterior height restoration and fusion of L5/S1 and L4/L5. A posterior revision surgery was not considered due to the wide decompression and obvious scar formation. For the correction of level L4/L5, an oblique anterolateral approach (OLIF) was selected due to considerable calcification of the aorta and the iliac vessels. At the L5/S1 level, a straight anterior approach was selected and an additional plate fixation (ATB) was performed.
At level L5/S1, a large cage with an angulation of 14 was selected and for L4/L5, a large cage with an angulation of 10 was placed. In order to perform a fusion, the cages were each filled with 6 mg of BMPII.
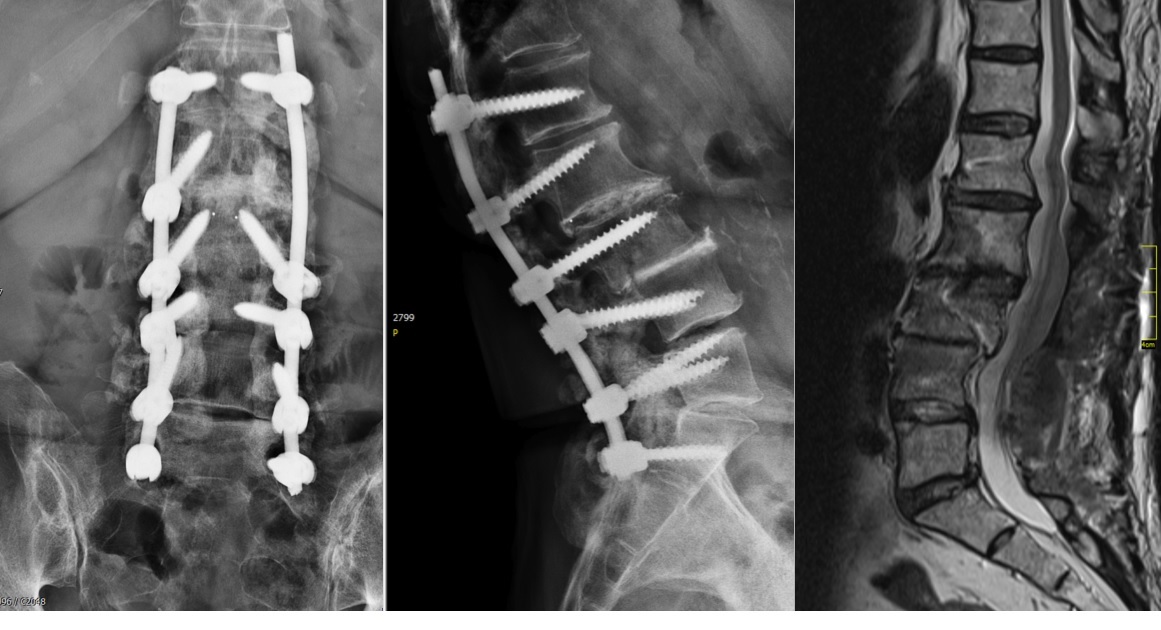
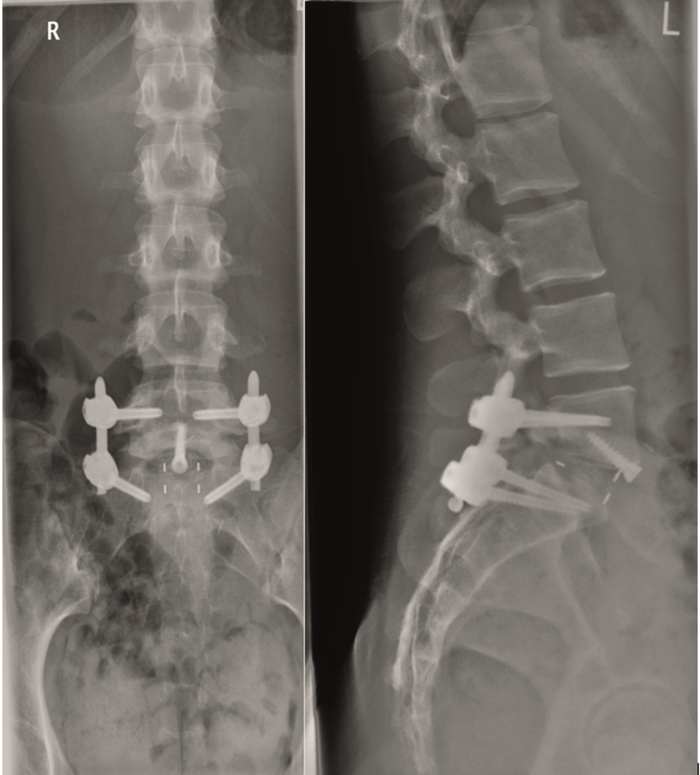
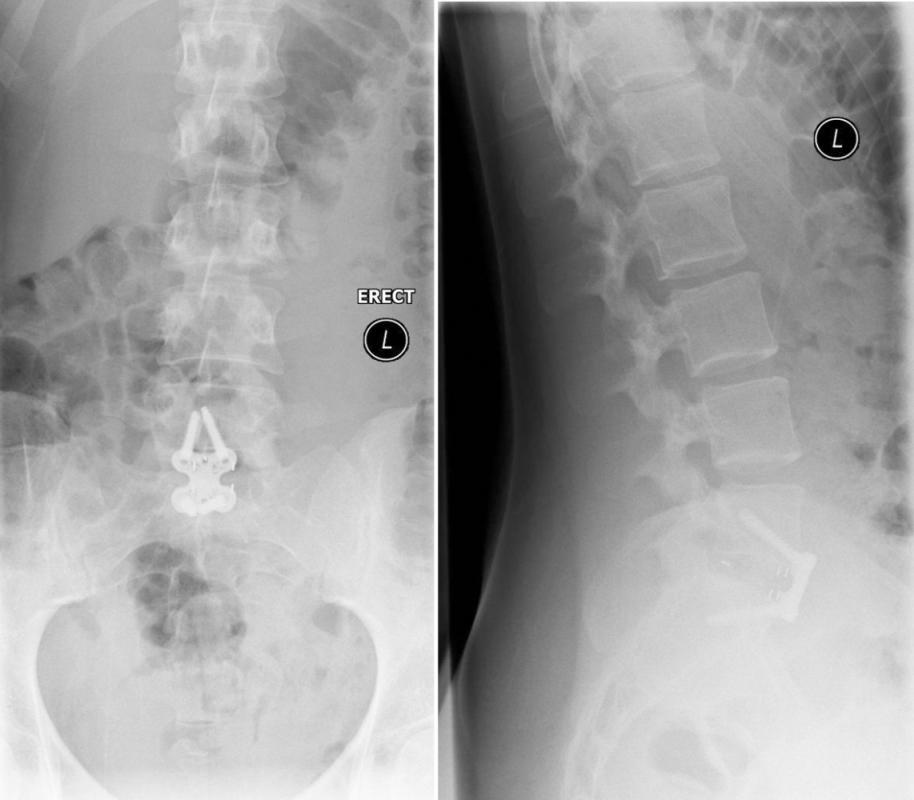
From six months postoperatively, leg discomfort decreased. Within an additional four months, pain disappeared completely and both foot and hip weakness recovered. The back pain persists to a certain extent but is not impedingthe patient in her daily activities. The x-ray taken 10 months after the anterior revision surgery revealed a complete and solid fusion on both levels (Fig 8). This is confirmed by the appearance of dense bone in the radiolucentcage.
Case provided by Khai Lam, London, UK
Case 2
A 19-year-old high-level college hockey player had experienced 12 months of severe lower back pain (LBP), and was unable to play sport due to high disability and pain (Fig 9). Nonoperative treatment with physiotherapy and injections had failed.
The CT showed bilateral L5 spondylolysis with grade I spondylolisthesis (Fig 10).
The patient underwent minimal access L5/S1 anterior interbody fusion with BMP followed by minimally invasive Matrix percutanous screw fixation (Fig 11).
Case provided by Khai Lam, London, UK
A 23-year-old female college student had experienced 3 years of severe LBP with some right S1 sciatica. She presented with high disability, failed nonoperative treatment, injections, and pain killers. She was unable to lead a normal life and conduct activities of daily living.
The sagittal and axial T2-weighted MRI showed grade III disc degeneration with diffuse right-sided disc bulging (Fig 12).
The postsurgery AP and lateral images show stand-alone locked L5/S1 anterior fusion using Syncage Evolution with BMP-2 and an Aegis locking plate (Fig 13).
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.