Locking Reconstruction Plate 2.4 and Mini Plate 2.0 System
This plating system offers a solution for mandible and maxilla fracture treatment in canines. The Mini Plate 2.0 is available in thicknesses of 1.0 mm/1.25 mm/1.5 mm/2.0 mm to address a variety of stability requirements. Furthermore, one screw size (2.0 mm) fits all Mini Plate 2.0 options (Fig 1) with the possibility of using a 2.4 mm cortex emergency screw when needed.
For larger dog breeds, the Locking Reconstruction Plate 2.4, with a thickness of 2.5 mm, should be the choice for fracture fixation in the mandible. This plate takes 2.4 mm and 3.0 mm locking screws, 2.4 mm cortex screws, and optional 2.7 mm locking cortex emergency screws (Fig 2). To fulfill the shape variation of the jawbones over the various dog breeds, the plates can be contoured in all three dimensions with appropriate instrumentation for perfect fitting. To prevent soft-tissue irritation, the plates have a low profile and highly polished surface.
Locking Design
The locking design increases the bone-plate construct stability by using angular stable screws. At the same time, it decreases the risk of screw back-out and subsequent loss of fracture reduction over time. The threaded Mini Plate holes accept both cortex and locking 2.0 mm PlusDrive screws. This configuration allows an angulation of the cortex screws of 13-18 from the central axis. The Reconstruction Locking Plate has a closely spaced hole-design pattern for optimized fracture fixation in minimal bone stock with locking screws or cortex screws.
Case: Cocker Spaniel
(Case provided by Randy Boudrieau, Massachusetts, USA)
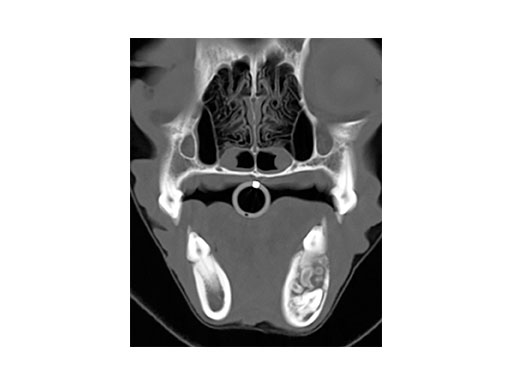
A 4-year-old castrated male cocker spaniel was admitted with a complex odontoma (confirmed by biopsy and histopathology) of the left mandible of 1.5 years duration. The CT image (Fig 3) shows the complex odontoma, and indicates an inhomogeneous mineral density within the mandible causing thinning/ disruption of the lateral cortex and tooth roots. The contiguous images demonstrated abnormal tissue spanning from PM3 to M1 of the left mandible.
Surgical reconstruction
A 12-hole Locking Reconstruction Plate 2.4 was contoured and secured to the ventrolateral mandibular border with three 3.0 mm locking screws inserted cranially and caudally. The most rostral 2 screws penetrated the canine tooth (a pulpectomy was first performed, since this is the only available point of screw purchase rostrally in the dog as the canine tooth fills the entire mandible at this location). The plate was then removed to facilitate resection of a 5 cm segment of the mandible from PM2 to M2 by transverse osteotomy between the teeth using an oscillating saw, obtaining 0.5-1 cm margins from the tumor. The plate was re-applied using the previous screw holes. The gingival margin was closed. An additional 16-hole, Mini Plate (intermediate size: 1.3 mm) was secured along the alveolar bone with four 2.0 mm locking screws cranially to the defect, and three 2.0 mm locking screws caudally. Two screws rostrally penetrated the canine tooth. The plate was anchored caudally to the coronoid crest of the ramus.
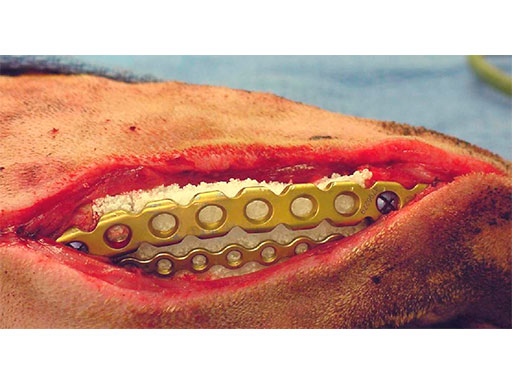
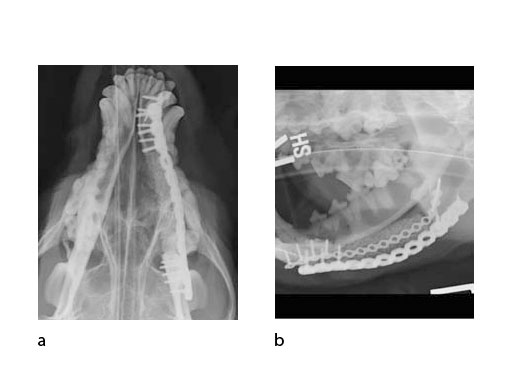
A block of compressive resistant matrix (CRM) was cut to fill this bone void, which was soaked with 2 mg of rhBMP-2. Fig 4 shows an intraoperative view of the 2 plates secured to the mandible and the CRM in place within the bone defect. The soft tissues were closed routinely. Immediate postoperative x-rays show the fixation (ventrodorsal view and lateral oblique view) (Fig 5). The radiopaque CRM can be observed spanning the 5 cm defect.
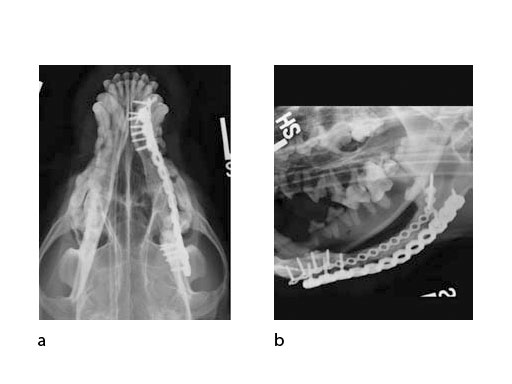
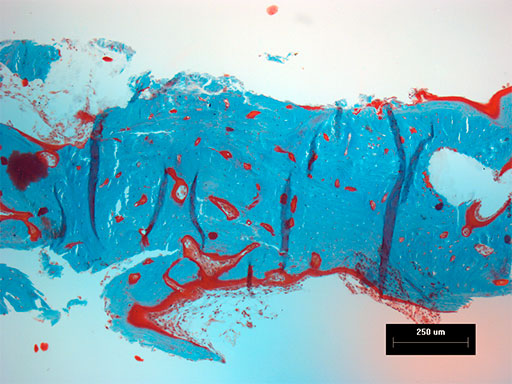
Follow-up x-rays at 7 months postoperatively (ventrodorsal view and lateral oblique view) (Fig 6) show healing of the defect (and resorption of the CRM), which was documented with a Jamshidi biopsy of the center of the original gap (Fig 7). The biopsy was performed at the same time as partial plate removal (Mini Plate) due to the intraoral plate exposure.
Long-term follow up of this dog was obtained in-hospital 26 months postoperatively (both plates were fully removed at 20 months postoperatively) (Fig 8), and via telephone follow-up 6 years postoperatively. At that time the dog was euthanized for an unrelated issue. However, the dog had done well with the mandibular reconstruction throughout this time frame.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.