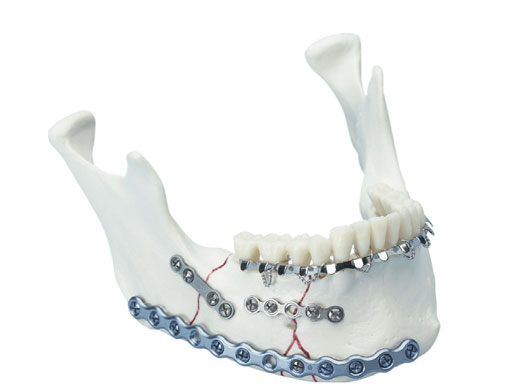
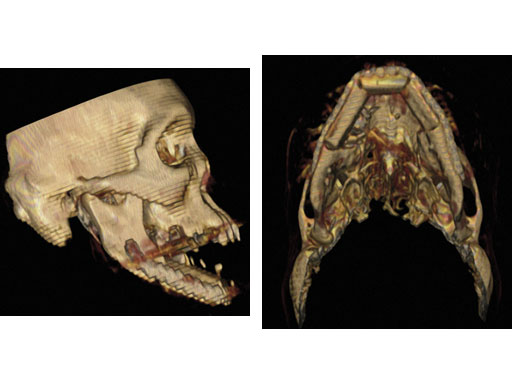
Matrix Mandible System
The new MatrixMANDIBLE System completes the family of existing craniomaxillofacial (CMF) Matrix Systems which are available for midface and neuro procedures, and orbital fracture repair.
The MatrixMANDIBLE set design is based on the well-received Mandible Modular Fixation System, and addresses surgical feedback and clinical need for a new consolidated set. It addresses the limitations and technical challenges for expanding into new technologies that could be seen in the old MLP/unilock 2.0 and LRP/unilock 2.4 systems due to their different designs and specific (noninterchangeable) instrumentation.
MatrixMANDIBLE 2.4 mm Cortex Screw Line Extension, April 2021
In April 2021, the MatrixMANDIBLE System was expanded with new 2.4 mm Titanium Cortex Screw in lengths of 20–40 mm. Read more in the "Details" tab below.
The new matrix mandible plate and screw system is intended for oral, maxillofacial surgery, trauma, reconstructive surgery, and orthognathic surgery related to the lower jaw, an anatomical structure which covers a fair amount of the daily procedures for CMF surgeons but also bears huge challenges for the developers of the system due to its complex biomechanical situation.
The main features of the system are:
- All screw diameters work with all plates
- A full set of new standardized instrumentation
- A single screwdriver blade can be used for all screws
- Improved screw/blade retention and reduced cam-out
- Off-axis screw insertion up to 15°
- Rounded soft-tissue friendly plate profiles
- Reduced inventory for hospitals without compromising clinical solutions
Plates
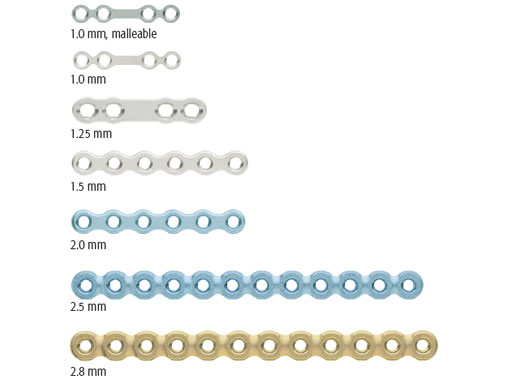
The plates in the system offer a conical locking technology for reliable screw-to-plate retention. All plates have rounded profiles and edges and an improved "angle plate design" to reduce stress in critical areas. The following plate thicknesses are available: 1.0, 1.25, and 1.5 mm (silver), and 2.0 mm (light blue) plates for trauma cases. (Malleable versions of the 1.0 mm plates are also available for load-sharing applications (color code: green-grey). For reconstructive purposes the range of plate thicknesses goes from 2.0 and 2.5 mm (light blue) to 2.8 mm (gold) with significantly improved fatigue strength. All plates are made from commercially pure titanium.
The 1.0 mm Thick, Adaption Plate is a shorter version of the existing 12 and 20 hole adaption plates. The bar in tension bands prevents the user from placing the screw too close to the fracture line, however, surgeon feedback indicated that 12 and 20 hole adaption plates were being used in a way where they were being cut into desired lengths. The fragmented plates were then used in different patients, which introduced challenging cost split. The new 4 and 6 hole adaption plates provides a more effective solution, and can be used without the bar as an option in trauma sets, allowing hospital savings.
The 2x2 hole 1.25 mm Thick, Tension Band is a stronger option for trauma and orthognathic surgery (BSSO) than the existing 1.25 mm Thick, Crescent Plate. It is 2 mm shorter than the 2x2 hole 1.0 mm Thick, Narrow Malleable Plate that is currently found in the set. Both the 1.25 mm Thick, Tension Band and its bending template have been added to the matrix mandible system.
Mandible reconstruction is at times necessary as a result of trauma or from segmental resection defects due to, eg, tumors. The main goals here are to restore functionality in terms of support mastication and swallowing, muscle attachment, maintenance of airways, and to provide a bed for teeth. It is also important to bear in mind aesthetic factors, such as the maintenance of facial contours and soft tissue.
In primary mandibular reconstruction with vascularized bone graft, it is often necessary to use lower profile plates. The 7x23 hole 1.5 mm Thick, Reconstruction Plate, left and right angle versions, and double angle in small, medium and large sizes have all been designed to improve concealment beneath the skin and reduce dehiscence in the mandibular region. Additionally, they can be used clinically in cases of comminuted mandibular fractures.
1. The 7x23 hole 1.5 mm Thick, Angle Reconstruction Plate, left side.
2. 1.5 mm Thick, Double Angle Reconstruction Plate small.
3. 1.5 mm Thick, Double Angle Reconstruction Plate medium.
4. 1.5 mm Thick, Double Angle Reconstruction Plate large.
Screws
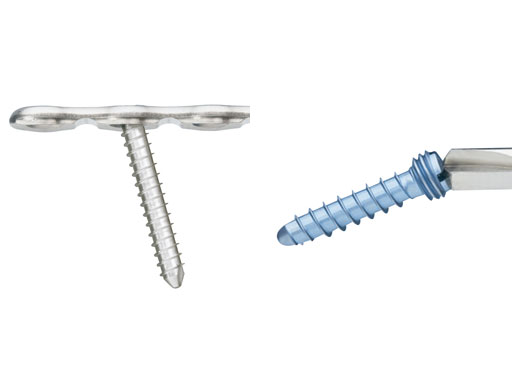
In keeping with the idea of the previously released matrix systems, all screw diameters in the set can be used with all the plates in the system. However, it is recommended to use those screws with the same color coding as the respective plate. 2.0, 2.4 and 2.9 mm diameter screws as well as 2.7 mm emergency (rescue) screws are available. All screws have a self-tapping tip with the 2.0 and 2.4 mm made available both with a locking or a nonlocking screw head design.
Depending on the size of the plate selected, a screw angulation of 10–15° and off-axis screw insertion up to 15° (screw-to-blade axis) can be achieved. All screws are made from titanium alloy.
Fig 3ab Screws.
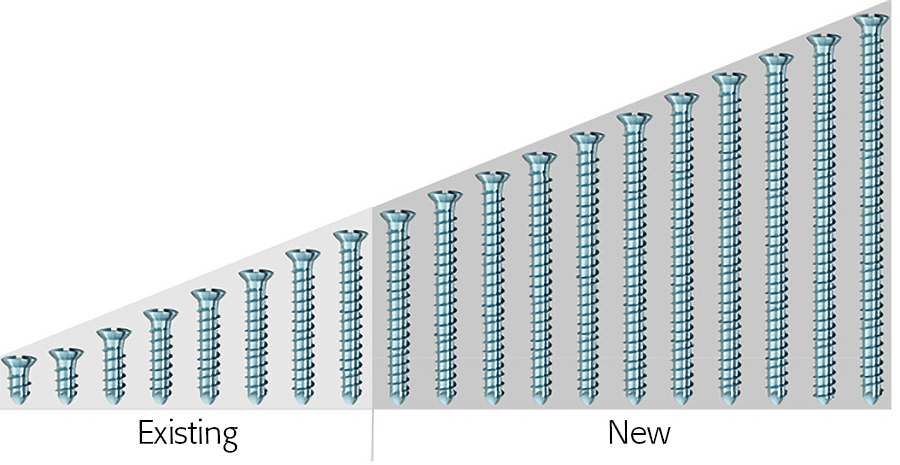
MatrixMANDIBLE 2.4 mm Cortex Screw Line Extension
In April 2021, the MatrixMANDIBLE System was expanded with the new 2.4 mm Titanium Cortex Screw in lengths of 20–40 mm.
The new 2.4 mm Titanium MatrixMANDIBLE Cortex Screw offers better flexibility for CMF surgeons in the treatment of mandibular fractures. Available in lengths of 20–40 mm, the new screw has the same design as the existing 2.4 mm Titanium MatrixMANDIBLE Cortex Screw (available in lengths of 5–18 mm). The existing 2.4 mm MatrixMANDIBLE Cortex Screw was found to be too short in certain surgical situations, for example when applied as a lag screw in the anterior mandible, or when used in oblique mandibular body fractures. In such situations, screws from the COMPACT System were often used instead. With the new additional screw lengths, all applications can be covered by one system.
The COMPACT Mandible System will be retired and superseded by the Matrix Midface System and the MatrixMANDIBLE System described above.
Instrumentation
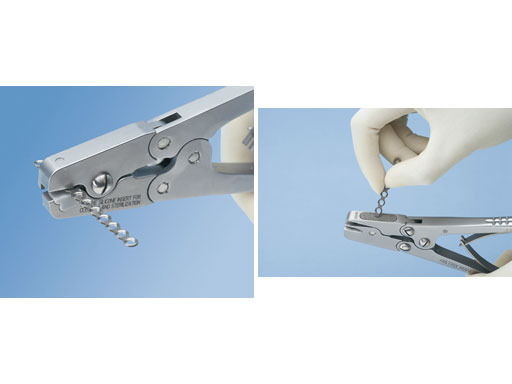
A specially designed set of instruments has been developed to achieve maximum efficacy with the implants in the system. Besides the feature that a single screwdriver blade can be used for all screws, new bending pliers for all plates and fixed-angle bending irons have also been developed. Furthermore diamond rasps have been added to the cutters to llow for easy on-site deburring of the cut edge of the plate.
Additionally available
The matrix mandible system will also provide the basis for future developments to continuously ensure state-of-the-art patient care. A few successful devices from older systems have been updated and integrated into the system already, ie, prebent tension plates for oblique line fractures and the condylar add-on system (see TK News 2/08). Another new plate which was based on the matrix mandible platform is the preformed (anatomical) reconstruction plate.
Fig 4ab Cutting pliers with deburr feature.
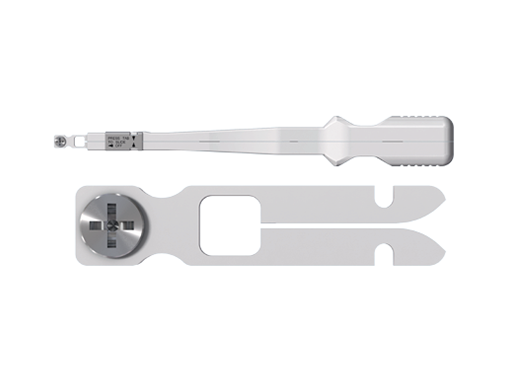
The Articulating Plate Introducer is a new version of an existing instrument that is only usable with dynamic compression plates (DCP) of the subcondylar/ramus fixation set. The tips provide versatility, since they are compatible with the matrix mandible 1.0 mm Thick, Straight Design and Subcondylar Plates, as well as 1.25 mm Thick, Non-DCP Plates. They are also interchangeable with the current instrument, so either the complete instrument or just the tips, can be optionally available for trauma treatment in conjunction with the transbuccal set.
1. Articulating Plate Introducer complete instrument.
2. Articulating Plate Introducer tip.
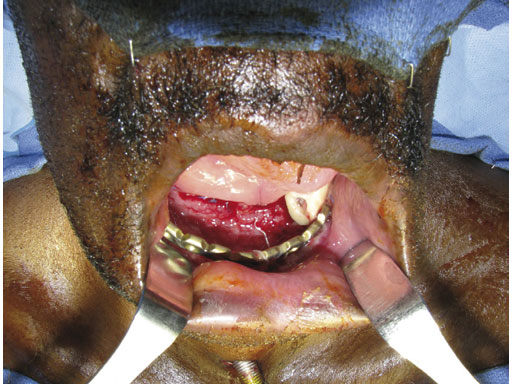
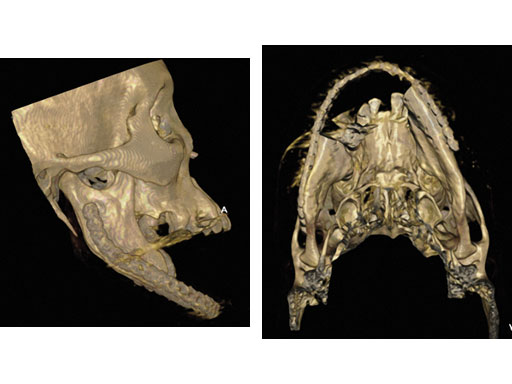
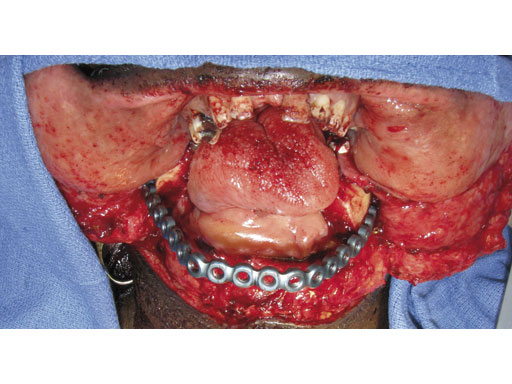
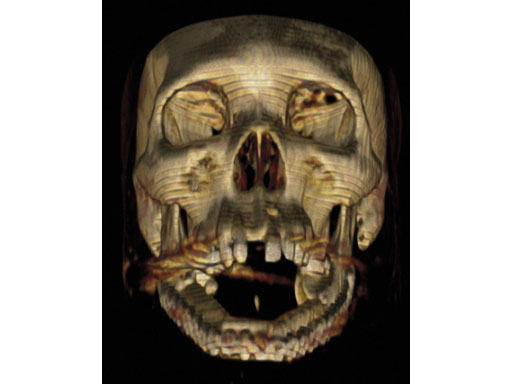
Case 1: A 47-year-old man transferred to the authors hospital approximately 10 days following the resection of a large, benign, locally aggressive odontogenic tumor of the mandible. He was primarily reconstructed with a locking reconstruction plate. The plate dehisced and the patient was unable to eat or swallow leading to his emergent admission to the authors hospital for reconstruction of his mandible with a composite vascularized flap (fibula). The initial internal hardware was removed and a 2.5 mm matrix mandible locking reconstruction plate contoured and applied to the remaining mandibular rami to aid in the reconstruction of the subtotal mandibular defect.
Fig 1 Intraoral situation showing exposed reconstruction plate and exposed edge of native mandible.
Fig 2 Preoperative situation.
Fig 3 ab Initial plate from lateral and submental vertex views.
Fig 4 After removal of the reconstruction plate, a new 2.5 mm mandible matrix reconstruction plate is contoured to restore the continuity of the mandible.
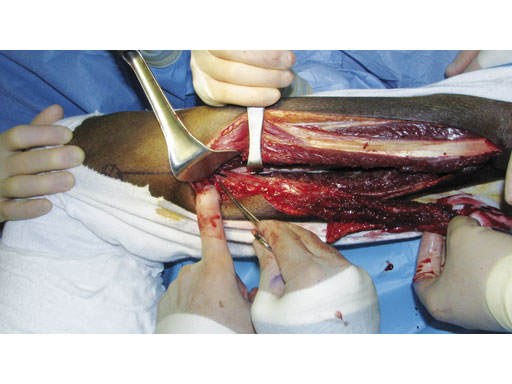
Fig 5 A fibula osteocutaneous flap is harvested to reconstruct the mandible.
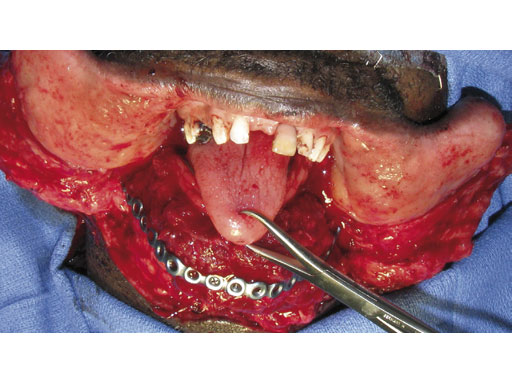
Fig 6 The fibula flap is contoured and inset to restore mandibular continuity and the skin paddle is used to restore the intraoral soft-tissue defect.
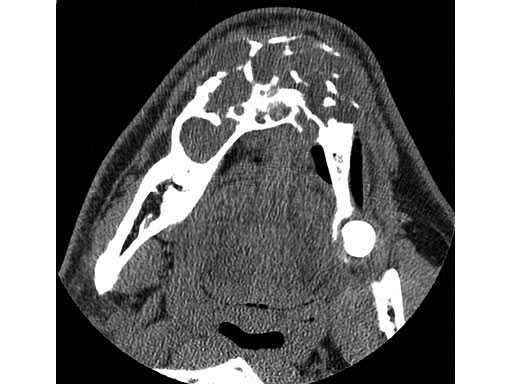
Fig 7ab Postoperative 3-D CT scans from lateral and submental vertex views.
Case provided by Daniel Buchbinder, New York, USA
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.