2.7/3.5 mm LCP Lateral Distal Fibula
Philip Kregor
The 2.7/3.5 mm LCP lateral distal fibula is an anatomically designed plate for the treatment of fractures, osteotomies, and nonunions of the metaphyseal and diaphyseal region of the distal fibula, especially in osteopenic bone.
The plate has an anatomically precontoured head and a low-profile design. It comes in right and left versions with lengths from 3 holes (73 mm) up to 15 holes (229 mm) and is available in stainless steel.
Metaphyseal fixation can be achieved by five coaxial distal locking holes in the plate head which use threaded interface to rigidly fix locking screws. These combination holes also accept standard cortex screws. Overall the plate head can either take 2.4 mm cortex screws, self-tapping (6-40 mm), 2.4 mm locking screws, self-tapping (6-30 mm), 2.7 mm cortex screws, self-tapping (6-55 mm), or 2.7 mm locking screws, selftapping (10-55 mm). A recess in the top of distal holes seats cortex screw heads to minimize cortex screw prominence. Also the locking screw heads are recessed. Four K-wire holes in the plate head accept 2.0 mm K-wires for provisional fixation.
For diaphyseal fixation the combination holes use the threaded side to hold locking screws at predetermined fixed angles, and the DCU side for conventional plating with nonlocking screws. An elongated combination hole holds the plate to the bone while allowing plate position adjustments. Plates with six or more holes have two elongated combination holes. The plate shaft accepts 3.5 mm locking screws, self-tapping (10-95 mm), 3.5 mm cortex screws, self-tapping (10-150 mm), and 4.0 mm cancellous screws with full or partial thread (10-100 mm).
The T8 and T15 StarDrive, 2.5 mm hex recess in the screws provides optimum torque transmission.
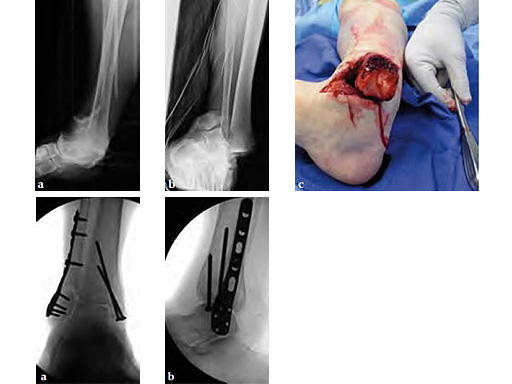
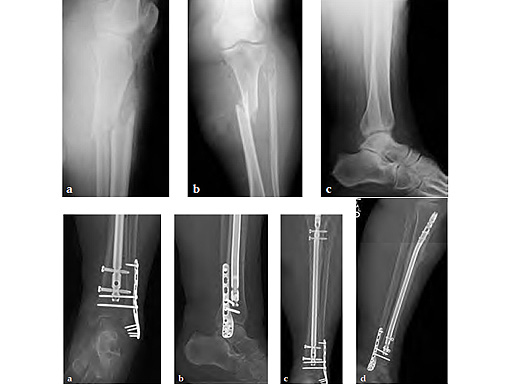
Cases provided by Mathew Graves, Jackson, USA.
Case 1: 55-year-old male fell off a horse and sustained an open pronation abduction ankle fracture dislocation.
Case 2: 80-year-old morbidly obese female. Post motor vehicle collision with open proximal third tibial shaft fracture, closed lateral malleolar fracture and fibular neck fracture, and distal tibiofibular syndesmotic disruption with ankle capsular injury.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.