2.4 mm VA -Locking Intercarpal Fusion System
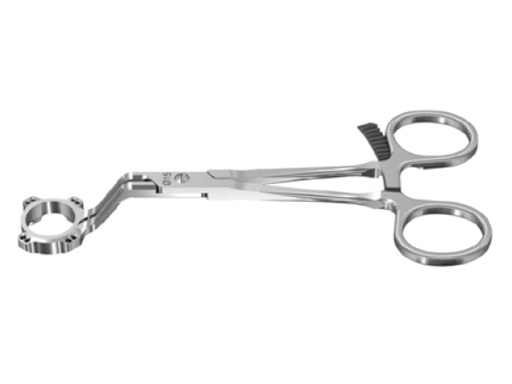
Fig 1: VA Locking Intercarpal Fusion Plate
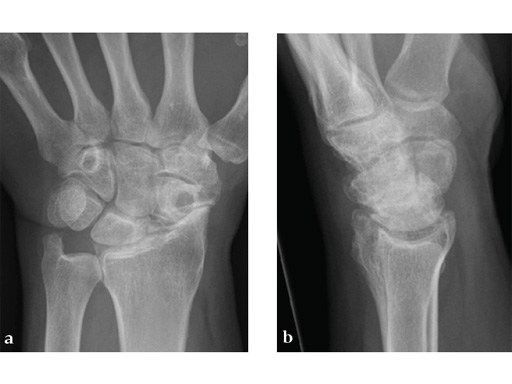
Midcarpal fusion involves excision of the scaphoid in order to address the painful radioscaphoid osteoarthritis. The midcarpal joint needs to be stabilized due to potential pain and arthritis depending on the stage of SLAC/SNAC. Stabilization is performed by arthrodesis of the lunate, capitate, hamate and triquetrum, subsequently named a 'four-corner fusion.' As the radiolunate joint is usually spared from arthritic changes, there is normally sufficient and pain-free residual motion between the lunate and the radius.
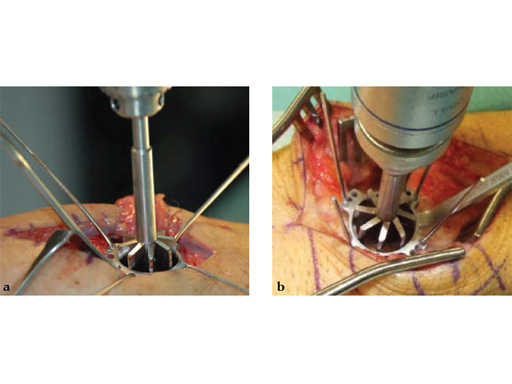
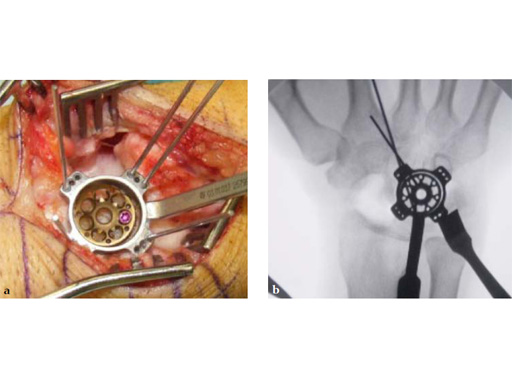
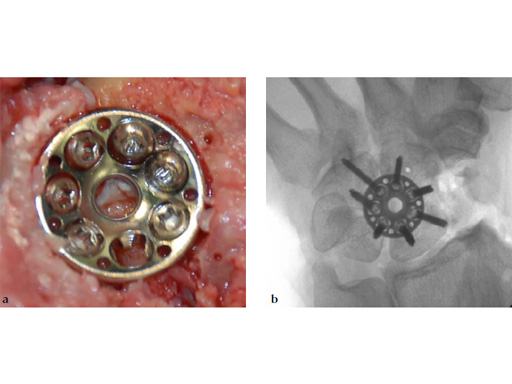
The 2.4 mm VA-locking intercarpal fusion system combines a circular plating technique with 2.4 mm variable angle locking technology and dedicated instrumentation . The circular plate design with a smooth surface and rounded edges minimizes soft-tissue irritation (Fig 2). To avoid impingement, proper positioning of the fusion as well as accurate placement and recession of the implant is mandatory. In addition to the standard reaming guide (Fig 3 in Instrument tab), the new reduction reaming guide permits reduction, compression and stabilization of the fusion site during the reaming procedure (Fig 4 in Instrument tab). Laser markings on the reamer support the surgeon by precisely controlling the depth of reaming for optimal countersinking of the plate (Fig 5 in Instrument tab).
The VA-locking plate holes accept both 2.4 mm cortex and VA locking screws. The use of cortex screws provides the option for compression by pulling the bone to the plate and locking screws enhance fixation in cancellous bone. The result is an extremely rigid construct that can withstand the strains of early postoperative mobilization. Midcarpal fusion provides a functional wrist motion achieving a minimum of 30° extension, 30° flexion, and 30° combined ulnar-radial deviation. This '30° rule' enables the patient to perform up to 90% of daily living activities.
The available two plate sizes include a 6-hole plate with a diameter of 15 mm, and a 7-hole plate with a diameter of 17 mm to fit different sized patients and applications.
Fig 3 Standard reaming guide with four pairs of K-wire fixation holes.
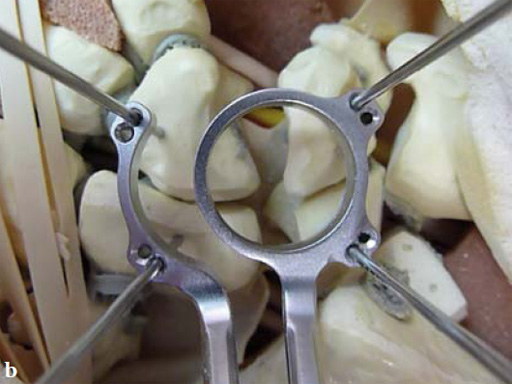
Fig 4a The reduction reaming guide also allows application of compression on the carpal bones.
Fig 4b The reduction reaming guide allows midcarpal distraction for bone graft packaging, and then application of compression at the carpal bones junctions.
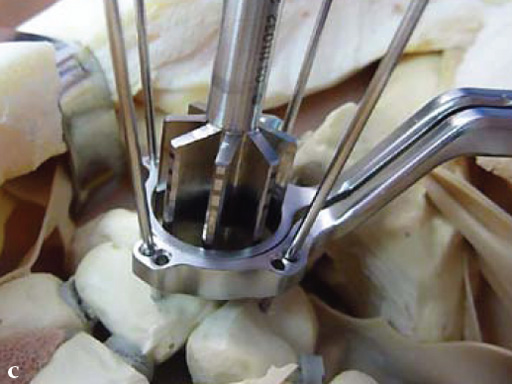
Fig 5 The reamer has 6 cutting teeth for the improvement of cutting performance. The laser markings ensure a safe and precise reaming depth.
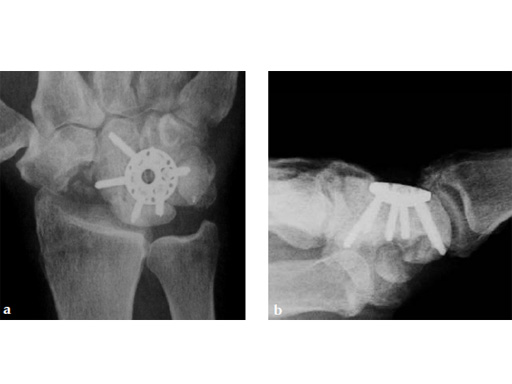
Case 1: SNAC wrist stage 3. Operative procedure and technical details.
Case provided by Ladislav Nagy, Zrich, Switzerland, and Fiesky Nez, Valencia, Venezuela.
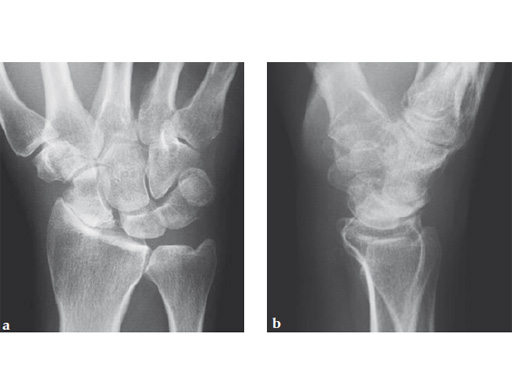
Fig 1ab Preoperative x-rays. Notice the midcarpal joint destruction and the DISI deformity of the lunate.
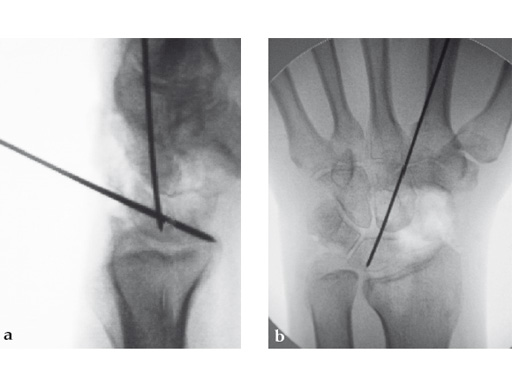
Fig 2ab Dorsal intercalated segment instability correction and K-wire fixation.
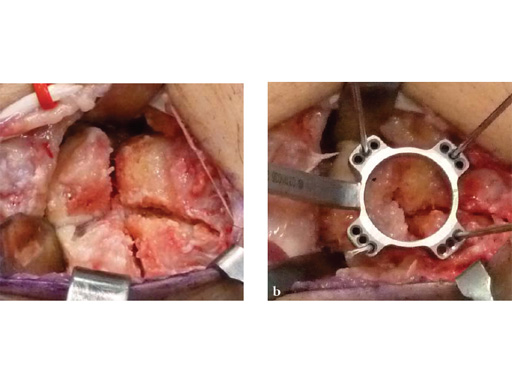
Fig 3ab Bone debridement. Guide positioning and fixation.
Fig 3c Bone debridement. Guide positioning and fixation.
Fig 4c Once the carpal junctions have been putting closer by the new reduction-compression guide, reaming is done.
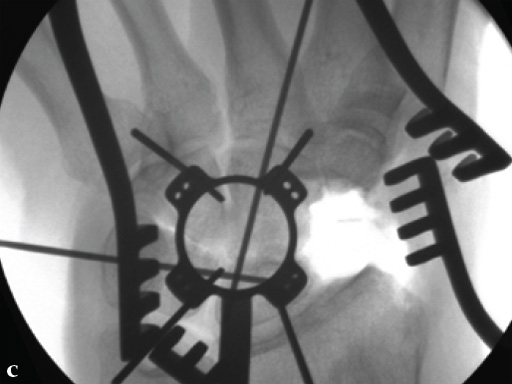
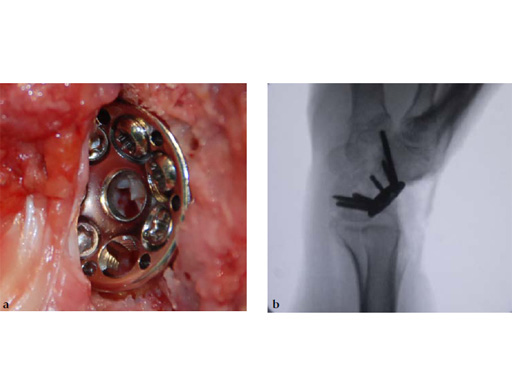
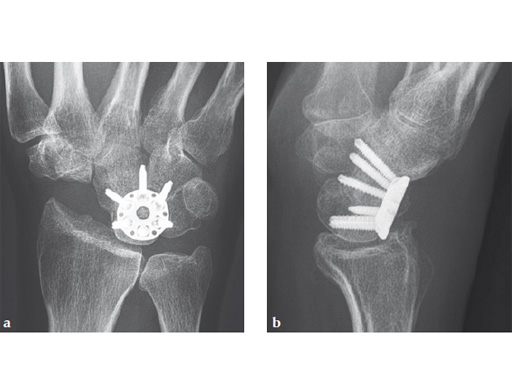
Fig 5ab Plate positioning and fixation. The combined VA-locking-standard guide allows drilling in the appropriate direction.
Fig 5cd Plate positioning and fixation. The combined VA-locking-standard guide allows drilling in the appropriate direction.
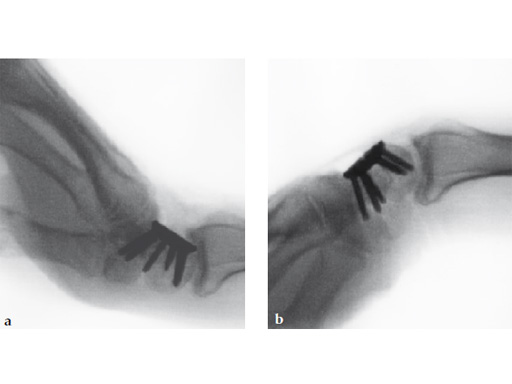
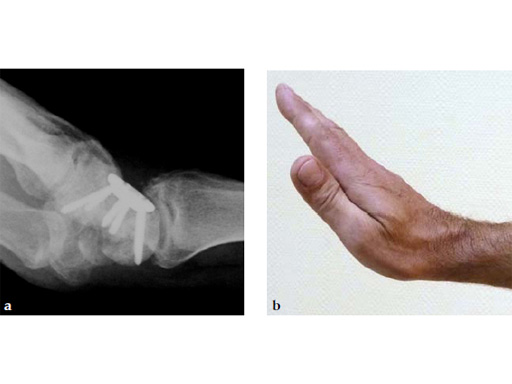
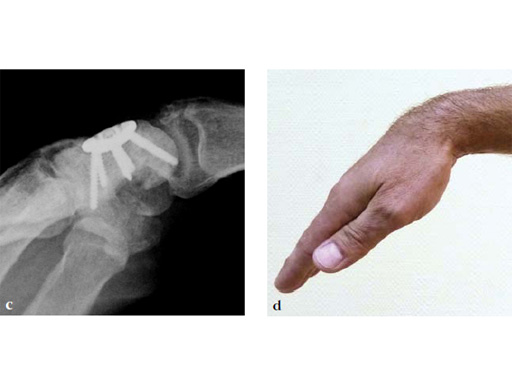
Fig 6ab Final appearance of fixed plate.
Fig 7ab No impingement of the plate against the dorsal rim of the radius in extension.
Case 2: Early degenerative wrist stage 3 secondary to a necrotic and collapsed proximal pole nonunion.
Case provided by Ariane Scheller, Berlin, Germany
Fig 1ab Preoperative x-rays. Note the carpal collapse a a b nd DISI deformity.
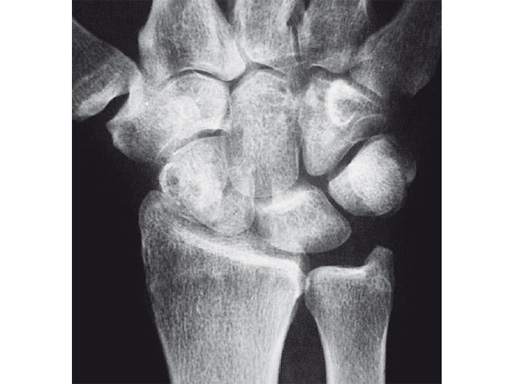
Case 3: Painful SNAC wrist in a 42-year-old man.
Case provided by Juan Gonzlez del Pino, Madrid, Spain
Fig 1 X-ray shows SNAC wrist.
Midcarpal Fusion Plate: The AO Four Corner Fusion Plate
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.