Expert Lateral Femoral Nail (LFN)
The Lateral Femoral Nail (LFN) allows a lateral entry site through the greater trochanter. The anatomic design is based on a femoral canal study. The helical shape has a 10 femoral anteversion.
The AO chose a lateral entry point because of the easy access to the entry site. This lowers the risk for an avascular necrosis in adolescents.
No splitting of the gluteus medius is needed and the damage of the gluteal medial tendon attachment minimized. Furthermore, the medial wall of the trochanter major is left intact.
The insertion point for the LFN is approximately 20 mm lateral to the center of the medullary canal, and 10 lateral to the greater trochanter, as measured from a point about 40 mm distal to the lesser trochanter. It is recommended to use the flexible reamer. The insertion follows the helical design: at the beginning the aiming arm shows anterior and turns continuously 90 helical to the lateral position at the end position.
The LFN is cannulated and will be available in two versions, either with standard and recon locking or with standard locking alone (only in US). The locking options have been enhanced. In proximal locking, the following options exist:
- 120 antegrade locking hole
- Transverse slot for either static or dynamic locking
- Two 6.5 mm recon locking holes (recon locking nail)
There are three distal locking options:
- Two transverse, lateral to medial holes
- One oblique distal locking hole offers enhanced stability of distal fractures
Indications for the LFN with standard locking are:
- Shaft fractures
- Impending pathologic fractures
- Nonunions, malunions
Indications for the LFN with recon locking are:
- Ipsilateral neck/shaft fractures
- Subtrochanteric fractures
- Shaft fractures
- Impending pathologic fractures
- Nonunions, malunions
The LFN is available in a left and right version with diameters from 916 mm and lengths from 300480 mm in 20 mm increments.
Cannulated, Stardrive end caps prevent ingrowth of soft tissue and facilitate nail extraction. The end caps can also be used to increase nail lengths.
The 5.0 mm and 6.0 mm standard locking screws feature a double-lead thread for ease of insertion. The thread is closer to the screw head which provides better bone purchase in the near cortex and improved
stability.
For the recon locking nail, 6.5 mm recon screws are available.
The LFN, as part of the expert nail family, allows the use of many instruments from the other systems such as drill sleeves, screwdrivers, depth gauges etc. The LFN has a color coding system to ease handling.
Expert Lateral Femoral Nail (LFN) study update
Intramedullary nailing is considered the method of choice in the treatment of femoral head and shaft fractures. Various nail implants are available for the management of these particular fractures. More recently, the lateral femoral nail (LFN) has been developed as part of the EXPERT nail family which offers an antegrade femoral nail with a unique anatomical bend for lateral entry point. The LFN is made with a different production technology, allowing for bending radii in two planes over portions of the nail. With an antecurvature of 1000 mm, LFN differs from other nails that have antecurvature characteristics of 1500 mm in the sagittal plane.
The LFN nail, with its two proximal locking options, (ie, retrograde (recon) and standard), is specifically indicated for the treatment of combined ipsilateral neck/shaft, subtrochanteric, and femoral shaft fractures with AO Classification 32-A, B, C and 31-B. With the recon locking option, two 6.5 mm screws can be inserted through the nail into the femoral neck and head. Alternatively, the LFN offers a standard proximal locking option with two 5.0 mm or 6.0 mm screws. Distal interlocking can be made by two static mediolateral, as well as one static oblique screw.
Due to advancements made with the LFN, AOCID has been conducting a prospective multicenter case-series that was initiated in August 2005, with the main objective of documenting all cases treated with this implant and evaluating intra-/postoperative complications associated with its use. Pain, fracture healing, reduction, functional outcome, and alignment were secondary parameters also monitored during the 12-week and 1-year follow-up periods.
17 study centers based in 11 different countries are participating in this prospective study. A total of 182 patients (130 male, 52 female) with femoral shaft fractures with or without femoral neck fractures have so far been recruited within a period of 17 months. The female patients are on average 20 years older than the male patients (32.2 male:51.8 female years), the general patient population is young (mean age:37.9 years) and consists mostly of fracture types 32-A3 and 32-B2, which have mainly resulted from traffic (ie, car/motorbike) accidents.
The LFN length and diameter most often used in this patient population is 360420 mm and 910 mm, respectively, and the average operation time of 118 min with an associated average x-ray exposure time of 341 sec is required for implantation. Recon locking option was used in 39/182 patients. Reaming occurred in 54% of the cases although for 84 unreamed cases, a small proportion (n = 14) of the nails was implanted with resistance. The occurrence of iatrogenic fractures (n = 6) was an observed baseline complication. During both follow-up examinations, other adverse events including namely screw/bolt breakage (n = 2) and delayed union (n = 7) or malrotation of the femur (n = 2) occurred.
The definitive evaluation of the study is expected to take place after the final follow-up assessments in spring 2008. This will provide us with the final results in regards to fracture healing, reduction, axial alignment, complications, reoperations, and evaluation on handling of the LFN.
Lateral insertion points in antegrade femoral nailing and their influence on femoral bone strains
Objective
Insertion of rigid uniplane bent femoral nails through the piriform fossa has been reported to cause muscular and neurovascular complications. New nails were designed for more lateral entry points. However, these may result in more iatrogenic fractures. This study investigated if two differently bent nails with more lateral entry points induce higher cortical bone strains than a uniplane bent nail introduced through the piriform fossa
Materials/methods
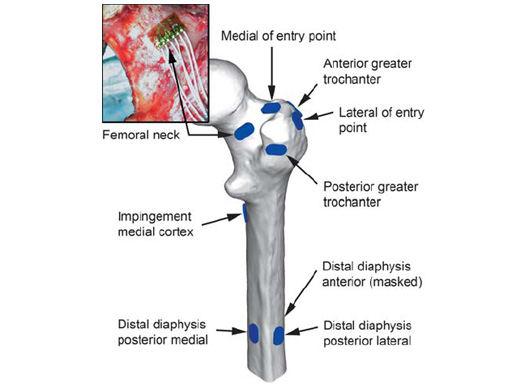
Three groups of eight cadaveric femurs were instrumented using following nail systems and entry points: cannulated femoral nail, piriform fossa (PF); antegrade femoral nail, trochanteric tip (TT); helical nail (prototype of the LFN), lateral of the trochanteric tip (LTT). During insertion the maximum principal bone strains were recorded at nine locations at the proximal femur and the diaphysis (Fig 1). The occurrence of iatrogenic fissures was documented.
Results
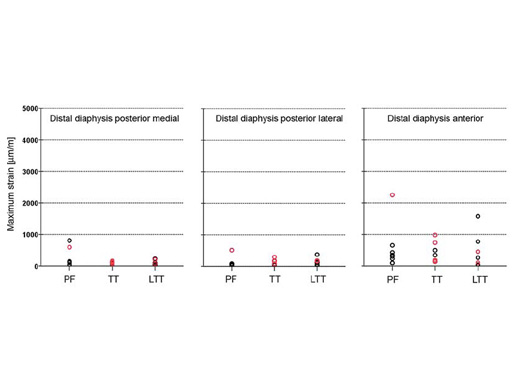
No differences in bone strains were found between the groups for the anatomical locations in the midshaft region and at the medial impingement point just below the lesser trochanter (Fig 2). Strains higher than those reported during walking, ie > 1800 m/m, were only recorded around the entry point. Significant differences between groups were observed at locations lateral to the entry point and the anterior greater trochanter, with the highest strains after nailing through the trochanteric tip. The high strains and associated fissures were due to deviations of the actually achieved insertion point from the ideal insertion point as planned from x-rays. Such deviations occured more often in the trochanteric tip group.
Conclusion
We conclude that nail introduction through entry points lateral to the piriform fossa does not induce higher bone strains distally and at the impingement site compared to the piriform fossa, when correspondingly designed nails are used and implanted in a correct way. For nails designed for an entry point at or lateral to the trochanteric tip, deviations of the actual insertion compared to the ideal insertion are associated with higher strains and sometimes fissures in the proximal area. However, the clinical relevance of these high proximal strains and fissures is unclear and subject to further investigation.
For soft tissue clearance in very obese patients, the standard insertion instruments may be too short, as the barrel of the standard insertion handle measures only 44 mm in length. Therefore, a longer barrel for the percutaneous insertion handle was designed with an increased length of 144 mm. The added length necessitates a longer mating connecting screw and a longer 5.0 mm hexagonal screwdriver to engage the LFN/R/AFN locking mechanism.
Because of its length a more proximal skin incision than is typically used is required. By concentrating on a more proximal skin incision in this obese population one might avoid the necessity of lengthening the skin incision and deep soft tissue dissection with the existing jig.
This additional handle should not be used on a normal nonobese patient as the added length of the arm may impinge on the soft tissues making the insertion more difficult.
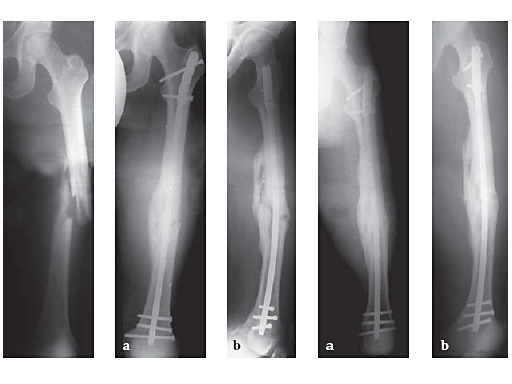
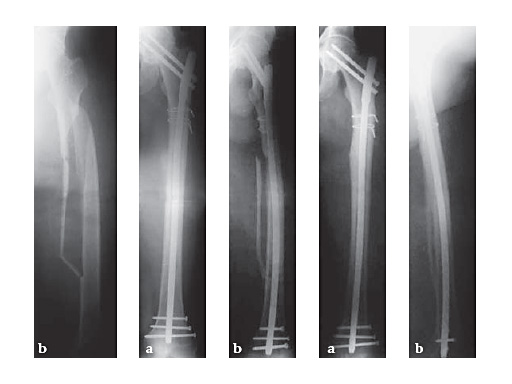
34-year-old malepolytraumatraffic accidentAO Classification 32-C3
Case provided by Hermann Bail, Berlin, DE
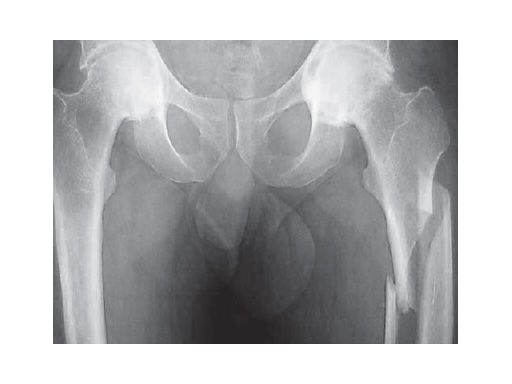
53-year-old malefallen in the busAO Classification 32-B1.1
Case provided by Hermann Bail, Berlin, DE
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.