Reamer-Irrigator- Aspirator (RIA)
The Reamer-Irrigator-Aspirator (RIA, Fig 1a) is indicated to clear the medullary canal of bone marrow and debris and to effectively size the medullary canal for the acceptance of an intramedullary implant or prosthesis.
The RIA provides irrigation, aspiration, and cooling during reaming, which allows single-pass reaming and reduced intramedullary pressure. Irrigation flows through the drive shaft and reamer head into the medullary canal. This irrigation mixes with the morselized medullary contents and is then removed through the aspiration tube.
The reamer heads range in size from 12 mm to 16.5 mm in diameter, in 0.5 mm increments. They are sterile packaged and intended for single-patient use. The reamer head is assembled to a disposable tube assembly which comes in two lengths, 360 mm and 520 mm. The tube assembly has a port for irrigation which is connected to saline solution via a standard irrigation set and an aspiration port which is connected to operation room suction using 1/4 suction tubing. The reamer head and the tube assembly are attached to a reusable drive shaft. A plastic seal prevents irrigation from flowing out of the system from the back end of the drive shaft and drive unit, which maximizes irrigation at the reamer head. Several investigations are ongoing to analyze the potential usage of the medullary contents.
In vivo assessment of the Reamer-Irrigator-Aspirator (RIA)
RIA vs unreamed nailing
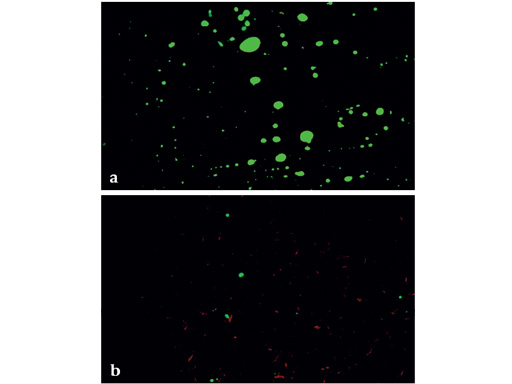
The purpose of this part of the study was to compare the effects of reamed intramedullary (IM) nailing using a new reaming system which uses irrigation and suction (RIA: Reamer-Irrigator-Aspirator; Synthes USA) with unreamed IM nailing; the goal being reduction of fat emboli in the blood, and coincidentally preventing occlusion of intracortical vessels. Sheep were treated with reamed (RIA) and unreamed (UR) intramedullary nailing on both unfractured femora. Blood samples were taken from both femoral veins during consecutive steps of IM nailing: UR: nail insertion; RIA: guide wire insertion, reaming, and nail insertion. They were assessed for fat content using a modified Gurd Test. Number and size of fat droplets was measured and data expressed as differences between nail insertion on the UR side and each step on the RIA side. In histological cross-sections intra-vital staining and fat staining, indicating functioning cortical perfusion and distribution of fat in cortical vessels, was evaluated. Ratios of perfused and fat containing vessels out of the total number of cortical vessels were then calculated.
Fracture healing was studied in the tibia. The procedure was first performed in the left tibia, with an observation period of ten weeks. Bone remodeling was labeled with Calcein green, Xylenol orange and Tetracycline. Pre-terminally the same nailing procedures were performed in the right tibia, followed by intravitam staining with Procion red, and an ink perfusion of the left tibia. The fractured left tibia was embedded in polymethylmethacrylate and histological sections and contact radiographs were evaluated with respect to callus development, cortical remodeling and ink perfusion. In the right tibia acute effects on cortical perfusion (Procion red staining) were evaluated in polymer embedded sections. Cryocuts were evaluated with respect to fat distribution (Sudan III staining).
Blood samples from the RIA side contained significantly less fat than the samples from the UR side (Fig 1b). After irrigation and suction little fat was observed in the cortex of RIA specimens, whereas after unreamed nailing the endosteal third of the cortical bone was infused with fat (Fig 2). Unreamed nailing acutely showed signs of remaining perfusion in the endosteal tenth and periosteal third of the cortical bone, after irrigation and suction-reaming perfusion was preserved to a lesser degree due to ablation (Fig 3). After ten weeks callus development and perfusion was similar in both groups. Cortical remodelling was the same in both groups after four weeks and was three times larger in the unreamed group after six weeks, indicating a more pronounced reaction to intracortical damage.
Irrigation and suction significantly reduces fat intravasation, and thus the danger of adverse systemic effects, while fracture healing is comparable to unreamed nailing.
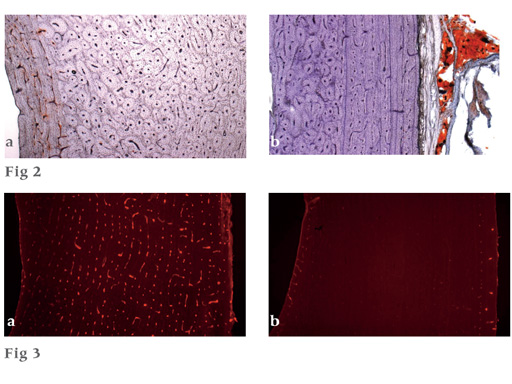
Upper pictures (Fig 2)
a Typical specimen of unreamed nailing with a red seam of stained medullary fat on the endosteal side of the cortex (left).
b Typical specimen of RIA nailing with no medullary fat visible in the cortex. The correct functioning of the staining technique is illustrated in the attached soft tissues on the right.
Lower pictures (Fig 3)
Perfusion did not show dramatic differences between the two nailing techniques, and disturbances covered a wide range.
a Example for a cortical region with relatively good blood supply.
b Example for an almost nonperfused cortical region.
RIA as a bone harvesting device
The RIA system permits the collection of bone particles which are produced by the reaming process. This system was used to study the influence of such bone particles, and of platelet rich plasma (PRP) on fracture healing. The results were compared to those of the SynReam (SYN) system.
In sheep, a defect of 8 mm was created in the left tibia at the midshaft. The RIA system was used in the groups RIA (no defect filling), RIA+B (bone particles) and RIA+B+PRP (bone particles+PRP), and the Syn-Ream system in group SYN. The medullary cavity was reamed to 11 mm in all four groups prior to implantation of an UHN (9.5 mm x 190 mm). After 6 weeks both tibia were harvested. Evaluation included mechanical testing, quantitative radiological evaluation and histomorphological analysis of newly formed bone.
For stiffness and strength the highest values were found in the group RIA+B, and they differed significantly (p<0.05) from group RIA. The values of RIA+B+PRP and RIA+B were significantly higher (p<0.01) than the values of SYN. After 4 weeks the radiological evaluation of callus area showed significant differences (p<0.05) between the two groups RIA+B+PRP and RIA+B and the two groups without bone reimplantation (RIA, SYN) but this difference was no longer visible at a later stage. No difference between RIA and SYN was found.
Callus formation can be significantly stimulated by the reimplantation of bone particles which are produced and collected by the RIA system. A clinical use of such techniques therefore seems to have an interesting potential.
Frequently Asked Questions about the RIA System
Annotated RIA Abstracts
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.