Locking X Plate
Post traumatic arthritis, rheumatoid arthritis, and neurological disorders can cause severe deformities such as cavus foot, equinovarus, telipes, and planovalgus. Treatment of these deformities by an osteotomy or a fusion of one or several joints bears the risk of screw breakage due to high shear forces. Other disadvantages are long healing and rehabilitation time. Metatarsophalangeal arthroplasty may leave a large defect if it fails.
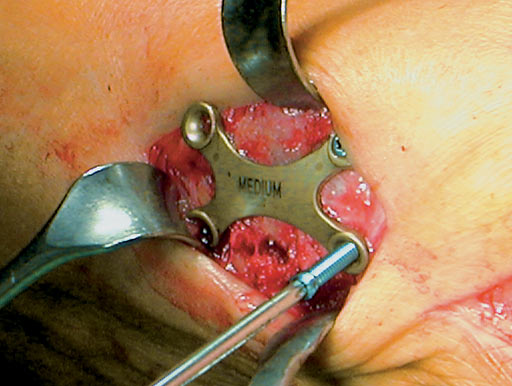
The Locking X-Plate 2.7 was developed to treat these deformities of the foot by a rigid fixation construct with high primary stability. It is a stand alone implant in osteotomies and serves as a neutralization plate in arthrodesis of foot joints in combination with one or two compression screws. Primary indications are first metatarso cuneiform fusions, proximal first metatarsal osteotomies (crescentic, open wedge, Mann, Ludloff, and proximal Chevron) and first metatarso phalangeal fusions.
The Locking X-Plate 2.7 is a geometric, low profile and easy to bend implant. This enables further usage for calcaneal osteotomies (Evans and Dwyer), metatarso nonunion fractures, supramalleolar osteotomies, and diabetic foot reconstruction.
Screw angulation prevents collision of the screws. The two dorsal screws are more angulated. The osteotomy can be crossed with a locking head screw when fixing a proximal metatarsal osteotomy providing high stability according to the locked internal fixator principle.
The X-Plate is fully compatible with the Compact Foot Set 2.7.
Now an additional size is available, extra small, with a foot print of 16 x 8 mm. Overall, there are four different sizes of the plates, according to the anatomical region and to the size of the foot: extra small, small, medium, and large. It is also likely to be used in countries where the ethnic anatomy is rather smaller than in other parts of the world.
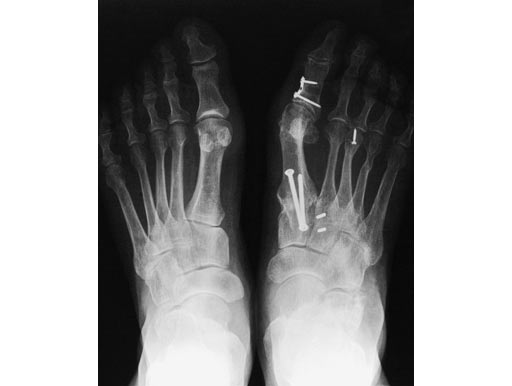
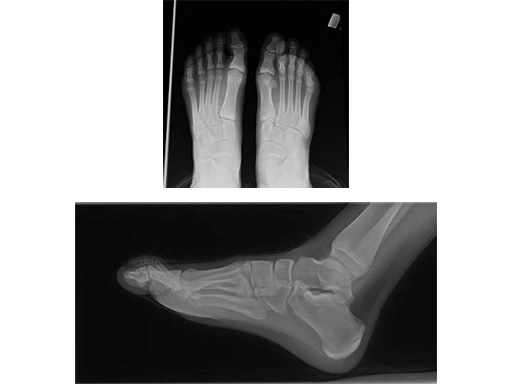
Case 1: 62-year-old female
One year after modified Lapidus operation with screws in the right foot. Now new treatment with an X-Plate (medium size) in the left foot.
Compared to screw osteosynthesis in the right foot, full weight bearing was possible after 6 weeks instead of 12 (see Fig 4)
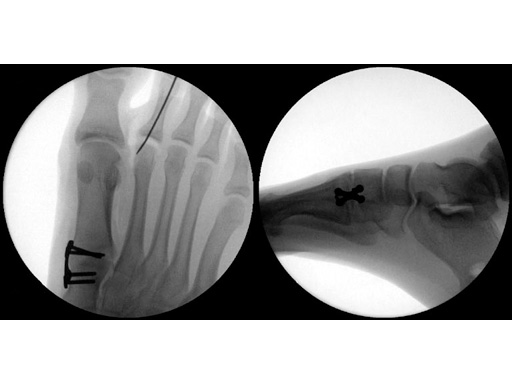
Case 2: 16-year-old male
Proximal first metatarsal growth-plate injury resulting in haluxvalgus. Arthrosis of first tarsometatarsal and second metatarsal hammer toe. Pain during activities and in general shoe wear.
Case provided by Carl Hasselman, Pennsylvania, USA
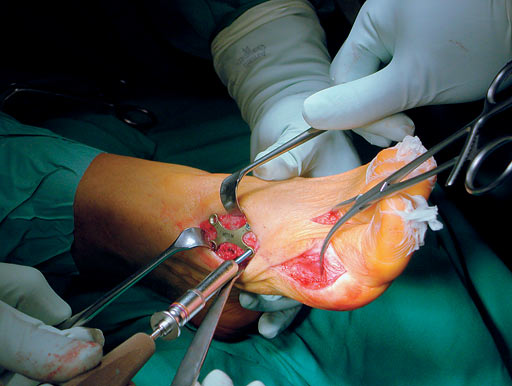
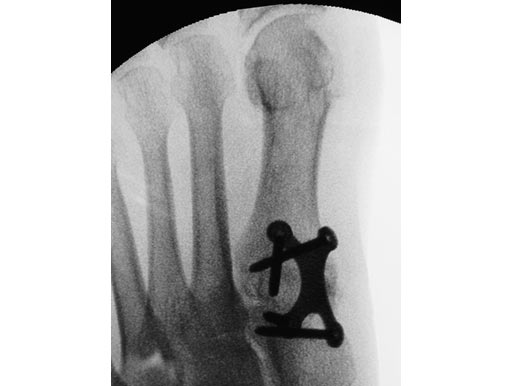
First MTP fusion and modified McBride with a distal softtissue release and second metatarsophalangeal (MTP) capsulotomy. A mini tight rope was used to hold and reduce the alignment of the first metatarsal. The X-plate was used to hold rigid fixation of the fusion. A K-wire was used for the second MTP capsulotomy.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.