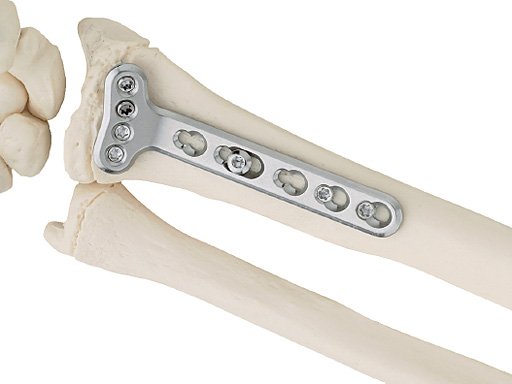
LCP Extraarticular Volar Plate 2.4, for distal radius
The LCP Extraarticular Volar Plate 2.4, for distal radius provides surgeons with a choice of a simpler fixation using extraarticular volar placement. It is indicated for fixation of complex intra- and extraarticular fractures and osteotomies of the distal radius and other small bones.
The plates are anatomically preshaped to fit on the volar side of the distal radius proximal to the articular surface. There are plates with either a 5-hole or 6-hole head, left and right. Shaft lengths can be chosen between either 3 or 5 holes.
The LCP Extraarticular Volar Plate 2.4, for the distal radius has a slightly different shape than the existing LCP Volar Plate for distal radius. It is wider and thicker (2.0 mm instead of 1.6 mm) which makes it stronger in bending.
The plate is available in stainless steel and titanium.
The short 1.8 mm threaded drill guide and insertion handles are instruments for use with the LCP Extraarticular Volar Distal Radius Plate, 4- and 6-hole versions.
The drill guides thread into all locking holes of all Volar Distal Radius Plates, or other plates where 2.4 mm and 2.7 mm locking head screws are used. The drill guide ensures that a screw hole is drilled at the correct angle in the head of an extraarticular plate. Multiple drill guides can be attached at one time in the head of the plate. They allow K-wire insertion. The drill guides are made from stainless steel and come with a T8 Stardrive recess.
The insertion handle is used with the short 1.8 mm threaded drill guides to position the Extraarticular Volar Distal Radius Plate correctly, and holds the plate in place while drilling. It is designed to keep the surgeons hand out of the field of view of the C-arm. The material of the handle is aluminum. 4-hole and 5-hole handles are available. One end of the handle is used with right plates and the other end is used with left plates.
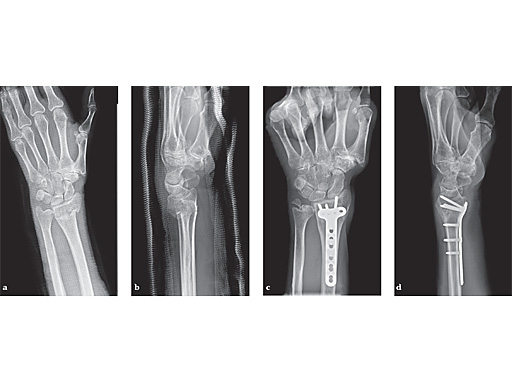
63-year-old female with skiing accident. Full motion and function was regained after 4 weeks.
ab Preoperative x-rays.
cd X-rays 6 months postoperatively.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.