Antegrade Femoral Nail (AFN)
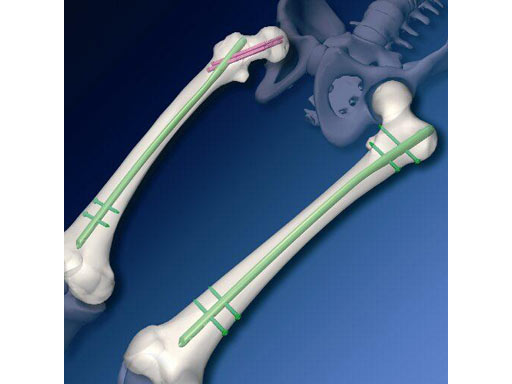
The Antegrade Femoral Nail (AFN) is an intramedullary device for treating femoral shaft fractures, as well as combinations of shaft fractures with subtrochanteric and femoral neck fractures approaching the medullary canal just lateral from the greater trochanter tip. The AFN is available from 9-14 mm in diameter. The proximal diameter varies according to the distal diameter. The core segment lengths range from 300 - 480 mm. The nail is cannulated with a 1500 mm radius in the sagittal plane, the average anatomical curvature of the femur. It has a lateral bend in the proximal part of 6 like the PFN. The AFN is manufactured from titanium alloy (TAN) in a right and left version, with three distal and four proximal locking holes. The AFN is generally compatible with the existing UFN/CFN instrumentation. Only a few additional instruments are needed.
Compared to the UFN/CFN, the AFN is advantageous due to the fact that:
- The anatomically pre-contoured nail facilitates insertion and provides a better fit into the medullary canal.
- The cannulation permits unreamed or reamed nail insertion over the central wire.
- The insertion point is easier to access at (or just lateral to) the tip of the greater trochanter. Additionally, it offers benefits regarding possible vascular or neurological lesions.
- The 6.5 mm hip screw ensures good hold in the femoral head and the enlarged screw provides compression.
- The cannulated endcap facilitates insertion using a guidewire with a hook.
The AFN is a new implant designed to stabilize femoral shaft fractures as well as combined fractures of the femoral neck and the femoral shaft. There are two main innovative changes to this nail compared with other AO approved implants used so far:
a) The material and form have been derived from the Proximal Femoral Nail (PFN). The entry point of this specifically curved titanium nail into the femoral cavity should, however, be located at the tip of the greater trochanter. This entry point has been chosen to ease the introduction of the nail and contrasts with the common use of straight femoral nails, like the Universal Femoral Nail (UFN), which in the majority of cases is inserted with more difficulty (especially in obese persons). As a cannulated nail, the AFN can be used in both reamed and unreamed techniques.
b) The AFN offers different proximal and distal locking options. With the new oblique proximal recon locking option, two 6.5 mm screws can be inserted through the nail into the femoral neck and head, thus offering the opportunity to also stabilize subtrochanteric fractures and combined fractures of the femoral neck and shaft. It therefore provides an alternative to more complicated procedures, eg, the miss a nail technique with the UFN. Alternatively, the AFN offers a proximal standard locking option with two transverse 4.9 mm pins. Distal interlocking can be carried out by two 4.9 mm pins, locked either in a static or dynamic fashion. The nail is available in several lengths ranging from 300 480 mm (20 mm steps) and three shaft diameters (1012 mm).
A prospective multicenter study has been conducted since October 2002 by AO CID to investigate the operative and postoperative complications associated with this new implant. Particular attention is being paid to complications connected with the entry point and the proximal recon locking option. The secondary objectives are to assess surgical details, such as the operative handling by performing surgeons, the anatomical restoration of the fracture, and the functional outcome of the patients.
In twelve teaching hospitals worldwide, 153 patients with femoral shaft fractures with or without additional femoral neck fractures were included in the study and treated with the AFN. Follow-up of the patients and prospective data collection will be performed until one year postoperatively. At each follow-up visit (six weeks, twelve weeks, one year) specifications about the patient, operative/postoperative complications, implant, recovery, and x-rays are documented. The preliminary analysis of the baseline data shows that no major technical problems occurred during nail implantation. The completion of the follow-ups is still ongoing until December 2004. The final report of the study results is planned for early 2005.
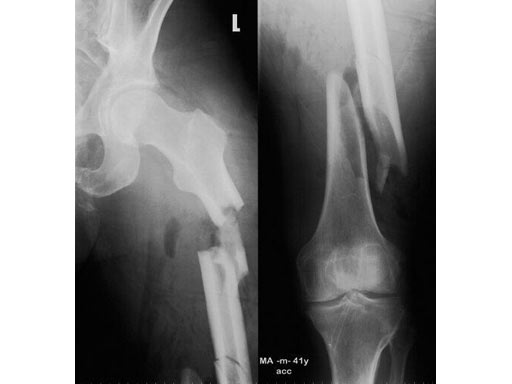
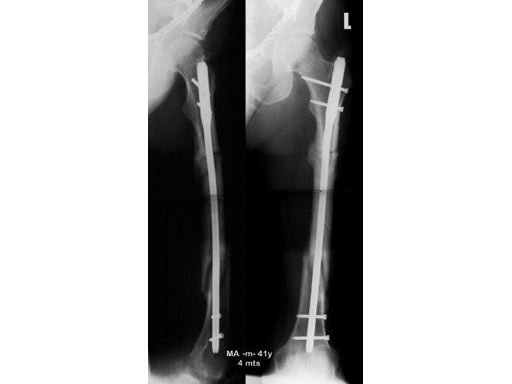
41- year- old male; accident with a motorbike.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.