VOLT™ Proximal Humerus Plating System
Simon Lambert, Frank Beeres, Joyce Koh, Stefaan Nijs, Martin Jaeger, Chunyan Jiang, Harry Hoyen
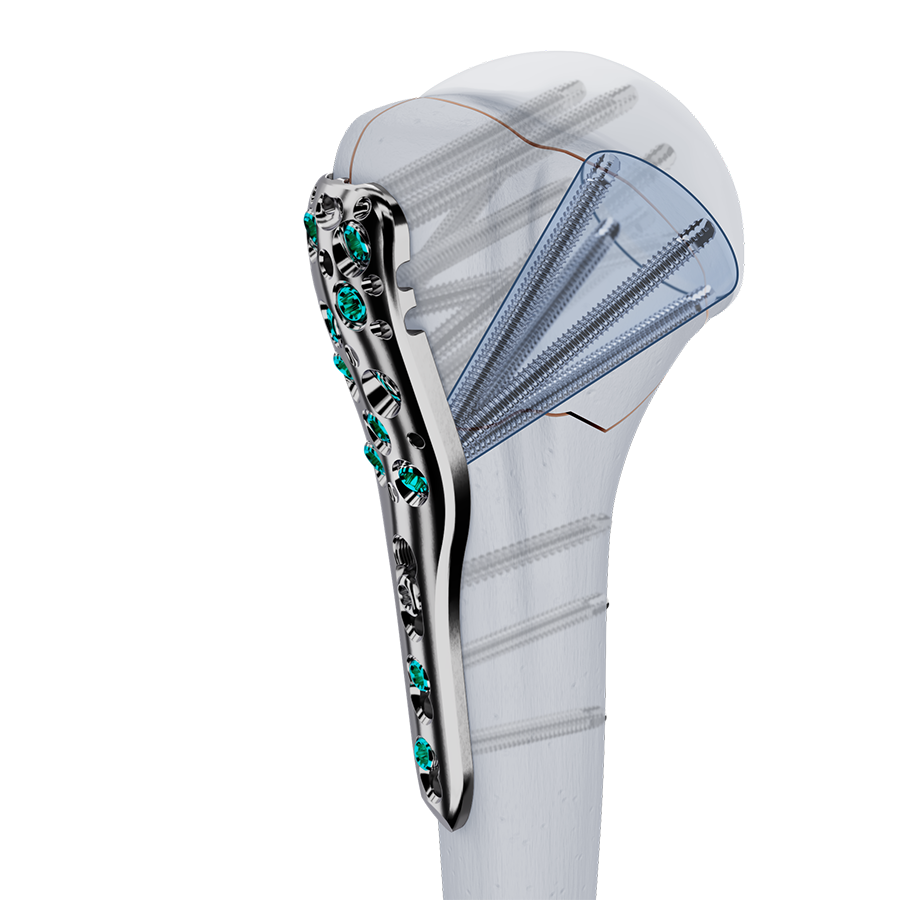
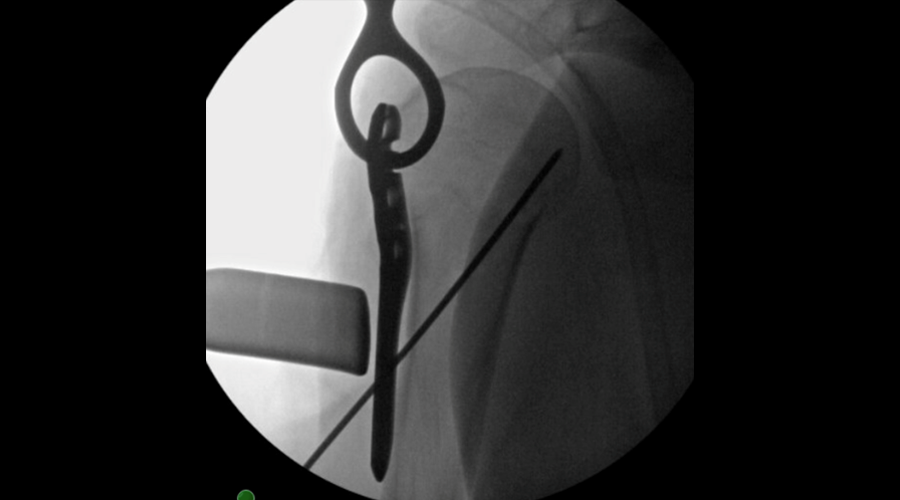
Approved by the AO Technical Commission in 2024, the VOLT™ Proximal Humerus Plating System addresses complex fractures of the proximal humerus. The newly designed plate holes feature locking with Variable Angle Optimized Locking Technology (VOLT™) which allows improved screw positioning (Fig 1) and improved flexibility in plate placement.
Enhanced design features of the plates include elongated combi holes which aid in plate positioning and accept both locking and cortex screws and limited-contact undercuts on the plate shaft. The new system features an expanded implant selection and offers a range of plate lengths to address a wider range of patients and fracture situations.
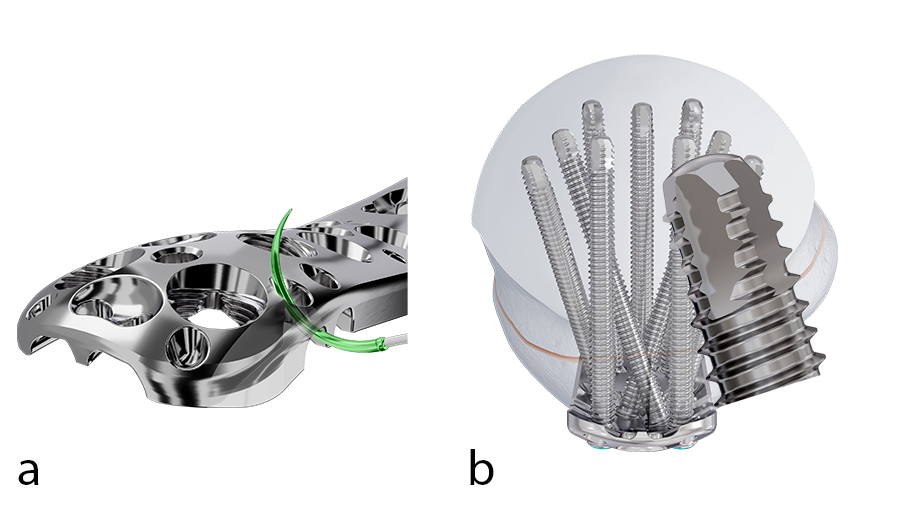
In the new plates, suture holes around the proximal end have been redesigned to allow the rotator cuff muscles to be more easily secured to the plate after implant insertion (Fig 2a). The system includes 3.5mm VOLT™ Locking Screws which feature an atraumatic tip (Fig 2b) designed to reduce the risk of soft tissue irritation compared to LCP.
Instrumentation has been improved with new, streamlined guide blocks addressing both deltopectoral and deltoid split approaches. A new length probe facilitates three different techniques for precise measuring.
System Overview: Plates and Screws
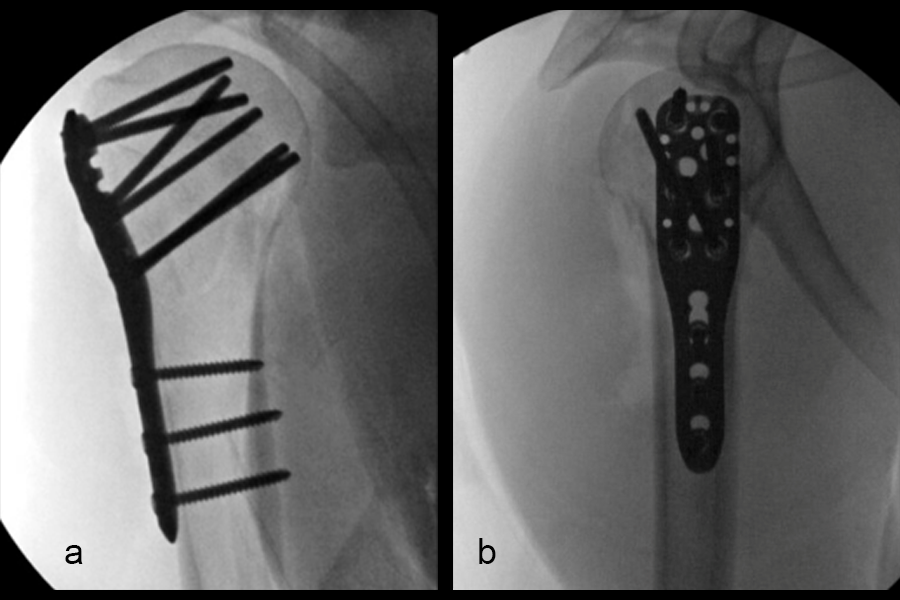
VOLT™ Proximal Humerus Plates are available in short (3 & 5 hole) and long (5, 6, 7, 8, 9, 10, 11, 12, 13 hole) versions (Fig 3). All plate lengths are available in stainless steel and titanium and are compatible with VOLT™ 3.5mm locking screws, 3.5mm cortex screws and 4.0mm cancellous screws. All implants feature 3.5mm VOLT™ locking and combi-holes.
Features and benefits
The design features of the new VOLT™ Proximal Humerus Plating System offer many benefits to both patients and surgeons:
- Precision locking with VOLT™ Variable Angle Locking Technology allows improved calcar screw positioning, more adjustability in plate placement - which is especially critical in smaller patients - and increased overall construct stability.
- The new length probe facilitates easier measuring of proximal screws and reduces the risk of screw penetration when bone quality is poor.
- The risk of interference with the axillary nerve is reduced when using the short guide block with the deltoid split approach.
- The new system is compatible with the VOLT™ Small Fragment platform of instruments with identical color-coding and a platform design approach. The ease of use facilitates training and adoption by surgeons and ORP, making instrument identification intuitive and reducing surgical delay.
Indications (US)
- The VOLT Proximal Humerus Plating 3.5 System is indicated for internal fixation of proximal humerus fractures in adults and adolescents (12-21 years) where growth plates have fused.
- VOLT Proximal Humerus Plates are indicated for fractures of the proximal end segment of the humerus.
- VOLT Proximal Humerus Plates, Long are indicated for fractures of the proximal end segment and/or diaphyseal segment of the humerus.
Indications (EU)
- VOLT Proximal Humerus Plates are indicated for internal fixation of fractures of the proximal end segment of the humerus.
- VOLT Proximal Humerus Plates, Long are indicated for internal fixation of fractures of the proximal end segment and/or diaphyseal segment of the humerus.
Contraindications
- No contraindication specific to these devices.
System Overview: Instruments
The VOLT™ Proximal Humerus Instruments include guide blocks, a length probe, a triple trocar, an outer sleeve for guided screw insertion, and 3- and 12-hole sizing templates. The streamlined guide block design is available in both a standard and short size (Fig 4), addressing both deltopectoral and deltoid split surgical approaches. The length probe offers versatility and precision with three measuring techniques: guide block (nominal angle), threaded drill guide (nominal angle) and locking drill guide (free hand, variable angle), depending on anatomic requirements.
Clinical Case: VOLT™ Proximal Humerus Plating System
Case kindly provided by Harry Hoyen, M.D. Metro Health Medical Center, Department of Orthopaedic Surgery, Cleveland Ohio, US.
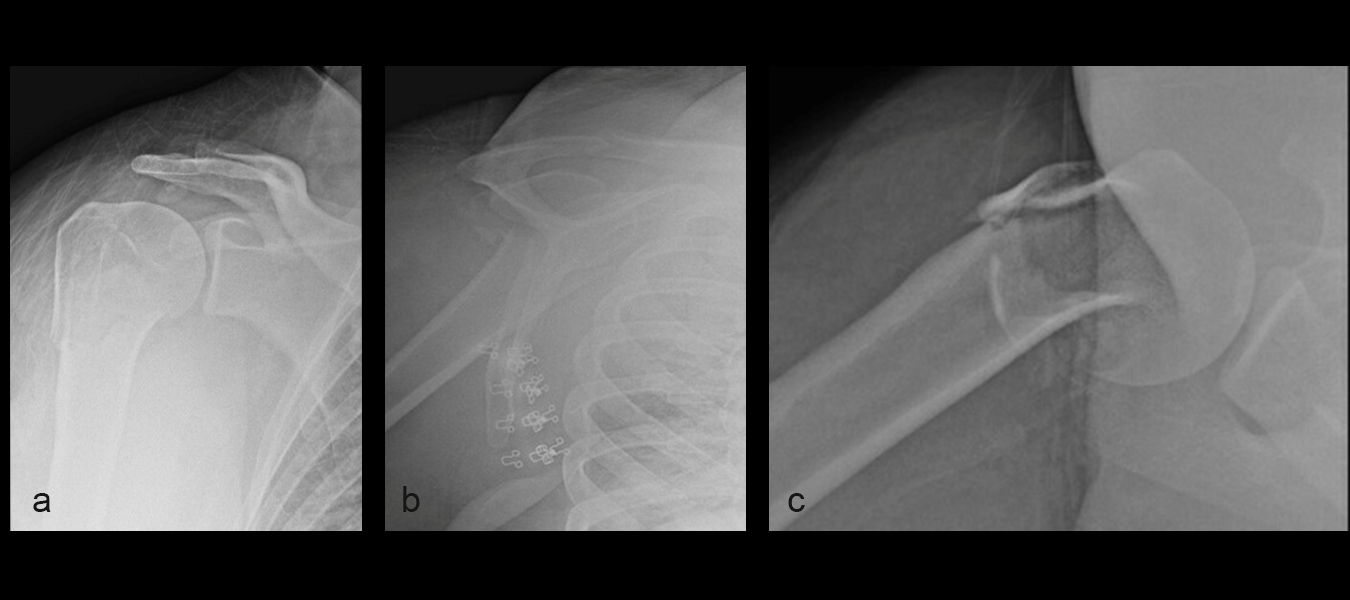
A 24-year-old woman presented to the emergency room following a motor vehicle accident. X-rays on the day of admission (Fig 1a-c) showed a fracture to the proximal humerus. There was some varus displacement of the humerus with involvement of the surgical neck and greater tuberosity.
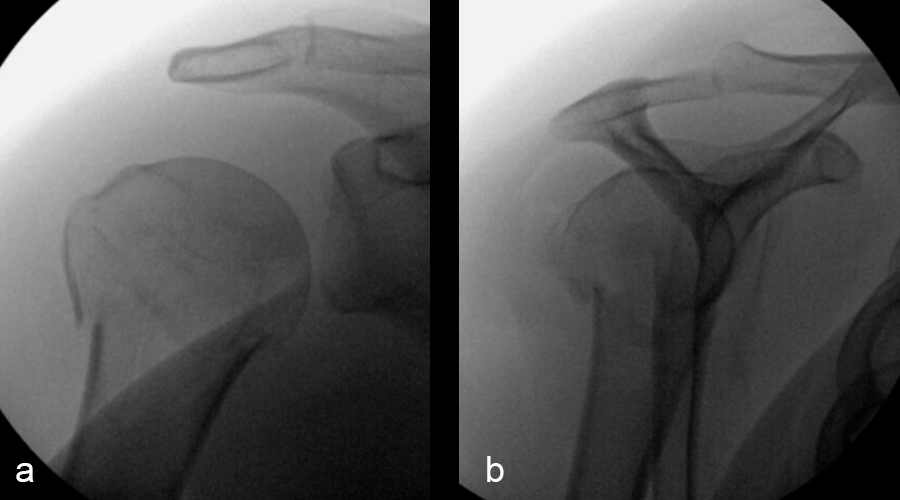
The patient was treated with open reduction and internal fixation (ORIF). First, the fracture was reduced (Fig 2a-b). There was an attempt at closed reduction with continued displacement. Then an open reduction through a deltopectoral approach was performed. After direct reduction, a 1.6 mm K wire was placed along the anterolateral humeral surface. The provisional plate placement and reduction are depicted in Fig 3.
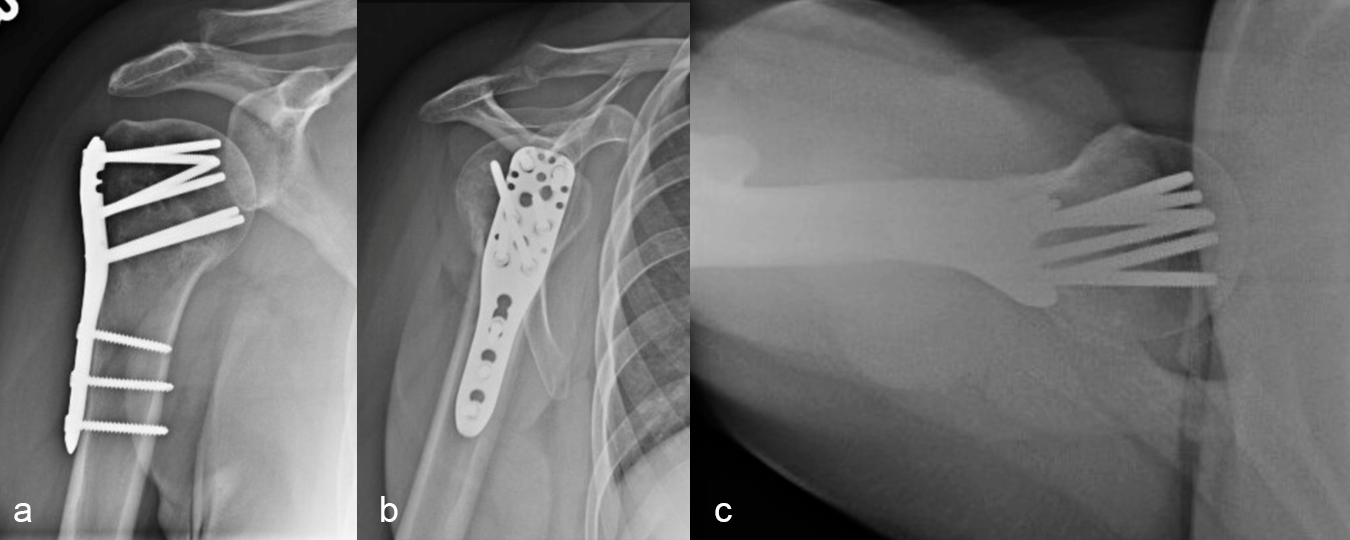
Definitive fixation was performed with a 3-hole VOLT™ Proximal Humerus plate (Fig 4). The plate was placed just beneath the rotator cuff insertion to directly support the greater tuberosity component. The proximal screws angulated toward the superior head to maximize length. The calcar screws were placed in a slightly splayed trajectory. One of the angular stable variable angled screws in the central portion was angulated in a posterior and more central position. This permitted maximal screw spread within the humeral head.
The patient was managed with sling immobilization for 2-3 weeks postoperatively. Early motion was permissible as the fracture was stable after fixation.
Three months after surgery, the patient had good elevation in the scapular plane to 130 degrees. External rotation was measured at 30 degrees, with more limited internal rotation (to the belt line). X-ray imaging (Fig 5) showed maintenance of fracture alignment and no change in screw position. The axial view (Fig 5c) highlights the subchrondral position of the variable angular screws in the humeral head. The patient was progressing nicely with functional tasks and adding weight in her therapy program.
VOLT™ Proximal Humerus Plating System
This session may contain graphic medical procedures and imagery intended for educational purposes. Viewer discretion is advised. The content is designed for healthcare professionals and may include sensitive or disturbing material.
Hazards and labeling
Due to varying countries’ legal and regulatory approval requirements, consult the appropriate local product labeling for approved intended use of the products described on this website. All devices on this website are approved by the AO Technical Commission. For logistical reasons, these devices may not be available in all countries worldwide at the date of publication.
Legal restrictions
This work was produced by AO Foundation, Switzerland. All rights reserved by AO Foundation. This publication, including all parts thereof, is legally protected by copyright.
Any use, exploitation or commercialization outside the narrow limits set forth by copyright legislation and the restrictions on use laid out below, without the publisher‘s consent, is illegal and liable to prosecution. This applies in particular to photostat reproduction, copying, scanning or duplication of any kind, translation, preparation of microfilms, electronic data processing, and storage such as making this publication available on Intranet or Internet.
Some of the products, names, instruments, treatments, logos, designs, etc referred to in this publication are also protected by patents, trademarks or by other intellectual property protection laws (eg, “AO” and the AO logo are subject to trademark applications/registrations) even though specific reference to this fact is not always made in the text. Therefore, the appearance of a name, instrument, etc without designation as proprietary is not to be construed as a representation by the publisher that it is in the public domain.
Restrictions on use: The rightful owner of an authorized copy of this work may use it for educational and research purposes only. Single images or illustrations may be copied for research or educational purposes only. The images or illustrations may not be altered in any way and need to carry the following statement of origin “Copyright by AO Foundation, Switzerland”.
Check www.aofoundation.org/disclaimer for more information.
If you have any comments or questions on the articles or the new devices, please do not hesitate to contact us.
“approved by AO Technical Commission” and “approved by AO”
The brands and labels “approved by AO Technical Commission” and “approved by AO”, particularly "AO" and the AO logo, are AO Foundation's intellectual property and subject to trademark applications and registrations, respectively. The use of these brands and labels is regulated by licensing agreements between AO Foundation and the producers of innovation products obliged to use such labels to declare the products as AO Technical Commission or AO Foundation approved solutions. Any unauthorized or inadequate use of these trademarks may be subject to legal action.
AO ITC Innovations Magazine
Find all issues of the AO ITC Innovations Magazine for download here.
Innovation Awards
Recognizing outstanding achievements in development and fostering excellence in surgical innovation.